Production and characterization of polyhydroxyalkanoates from seaweed associated bacteria
*Article not assigned to an issue yet
Alagarsamy Atsaya, Rajendiran Ramya, Kooloth Anju, Pasupathi Mukil Sukitha, Velu Rajesh Kannan
Research Articles | Published: 20 April, 2026
First Page: 0
Last Page: 0
Views: 74
Keywords: Polyhydroxyalkanoates (PHAs), Biopolymers, Seaweed-associated bacteria, Biodegradation, FT-IR and SEM characterization
Abstract
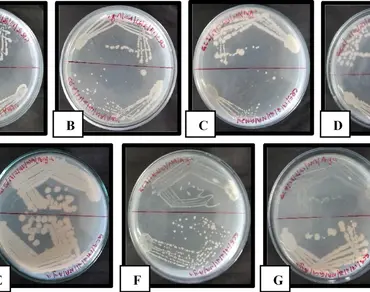
Plastics, derived from petrochemicals, are a type of resistant compound extensively used in agriculture, medicine, and various other sectors. Their inability to decompose poses a significant environmental threat. Consequently, biopolymers produced by microbes can serve as alternatives to synthetic plastics. Polyhydroxyalkanoates (PHAs) are biodegradable polymers created by different bacteria from substrates like sugars and fatty acids. This study aimed to isolate and characterize PHA–producing bacteria from seaweed and to evaluate their potential for biodegrading selected polycyclic aromatic hydrocarbons (PAHs). In this research, we gathered seaweed samples and isolated the bacteria associated with them. The ability of these bacteria to produce PHA was assessed using Sudan Black B staining, and their efficiency was evaluated by examining growth in a PHA production medium. The molecular identification of the efficient strain was identified as Bacillus tequilensis (CR4). PHA production was optimized using glucose and molasses as carbon sources, and the resulting PHA was characterized through FT-IR and SEM analysis. Additionally, this strain demonstrated the ability to degrade PAHs, indicating metabolic versatility. However, these findings suggest that seaweed-associated PHA-producing bacteria possess dual potential for biopolymer production and environmental bioremediation. This study is limited by the lack of large-scale production analysis, and future research focuses on genetic characterization of PHA producing bacteria and process optimization for industrial scale production.
References
Chaudhry WN, Jamil N, Ali I, Ayaz MH, Hasnain S (2011) Screening for polyhydroxyalkanoate (PHA)-producing bacterial strains and comparison of PHA production from various inexpensive carbon sources. Ann Microbiol 61(3):623–629. https://doi.org/10.1007/s13213-010-0181-6
Doley R, Barthakur M, Goswami BS (2017) Microbial degradation of aromatic hydrocarbon: naphthalene through Nocardiopsis alba RD3. Int J Curr Microbiol App Sci 6:1174–1181. https://doi.org/10.20546/ijcmas.2017.607.142
Duraimurugan R, Santhosh S, Mohamed Hanifa SH, Rao TN, Narenkumar J, Fang Z, Devanesan S, Wadaan MA, Rajasekar A, Rajamohan R (2026) Elucidating the mechanisms of anthracene and phenanthrene degradation by the halophile Vreelandella piezotolerant DM1. Extremophiles 30(1):1. https://doi.org/10.1007/s00792-025-01414-1
Edo GI, Ndudi W, Ali AB, Yousif E, Jikah AN, Isoje EF, Ali ABM, Igbuku UA, Mafe AN, Opiti RA, Madueke CJ, Essaghah AEA, Ahmed DS, Umar H (2025) Biopolymers: an inclusive review. Hybrid Adv. https://doi.org/10.1016/j.hybadv.2025.100418
Gregory DA, Taylor CS, Fricker AT, Asare E, Tetali SS, Haycock JW, Roy I (2022) Polyhydroxyalkanoates and their advances for biomedical applications. Trends Mol Med 28(4):331–342. https://doi.org/10.1016/j.molmed.2022.01.007
Hussain T, Roohi A, Munir S, Ahmed I, Khan J, Edel-Hermann V, Tassadaq H, Aneela R, Shehzad M, Iftikhar A, Jafar K, Veronique EH, Kil YK, Muhammad A, Anees M (2013) Biochemical characterization and identification of bacterial strains isolated from drinking water sources of Kohat, Pakistan. Afr J Microbiol Res 7(16):1579–1590. https://doi.org/10.5897/AJMR12.2204
Ibrahim R, Aranjani JM, Prasanna N, Biswas A, Gayam PKR (2025) Production, isolation, optimization, and characterization of microbial PHA from Bacillus australimaris. Sci Rep 15(1):8395. https://doi.org/10.1038/s41598-025-92146-x
Jiang Y, Song X, Gong L, Li P, Dai C, Shao W (2008) High poly (β hydroxybutyrate) production by Pseudomonas fluorescens A2a5 from inexpensive substrates. Enzyme Microb Technol 42(2):167–172. https://doi.org/10.1016/j.enzmictec.2007.09.003
Kumar M, Rathour R, Singh R, Sun Y, Pandey A, Gnansounou E, Andrew Lin K-Y, Tsang DCW, Thakur IS (2020) Bacterial polyhydroxyalkanoates: opportunities, challenges, and prospects. J Clean Prod 263:121500. https://doi.org/10.1016/j.jclepro.2020.121500
Możejko-Ciesielska J, Kiewisz R (2016) Bacterial polyhydroxyalkanoates: Still fabulous? Microbiol Res 192:271–282. https://doi.org/10.1016/j.micres.2016.07.010
Munir S, Jamil N (2018) Polyhydroxyalkanoates (PHA) production in bacterial co-culture using glucose and volatile fatty acids as carbon source. J Basic Microbiol 58(3):247–254. https://doi.org/10.1002/jobm.201700276
Nahar S, Jeong MH, Hur JS (2019) Lichen-associated bacterium, a novel bioresource of polyhydroxyalkanoate (PHA) production and simultaneous degradation of naphthalene and anthracene. J Microbiol Biotechnol 29(1):79–90. https://doi.org/10.4014/jmb.1808.08037
Odeniyi OA, Adeola OJ (2017) Production and characterization of polyhydroxyalkanoic acid from Bacillus thuringiensis using different carbon substrates. Int J Biol Macromol 104:407–413. https://doi.org/10.1016/j.ijbiomac.2017.06.041
Pauter K, Railean-Plugaru V, Złoch M, Pomastowski P, Szultka-Młyńska M, Buszewski B (2022) Identification, structure and characterization of Bacillus tequilensis biofilm with the use of electrophoresis and complementary approaches. J Clin Med 11(3):722. https://doi.org/10.3390/jcm11030722
Ramya R, Manikandan A, Rajesh Kannan V (2017) Standardization of biopolymer production from seaweed associative bacteria. Int J Biol Macromol 102:550–564. https://doi.org/10.1016/j.ijbiomac.2017.04.032
Sachan RSK, Kumar A, Karnwal A, Paramasivam P, Agrawal A, Ayanie AG (2025) Screening and characterization of PHA producing bacteria from sewage water identifying Bacillus paranthracis RSKS-3 for bioplastic production. BMC Microbiol 25(1):136. https://doi.org/10.1186/s12866-025-03841-8
Salam MA, Hussain A, Uzma, Bogale A, Iqbal Z (2025) Isolation of high yield polyhydroxyalkanoate producing bacteria from contaminated soils and biopolymer characterization. Sci Rep. https://doi.org/10.1038/s41598-025-29352-0
Samorì C, Kiwan A, Torri C, Conti R, Galletti P, Tagliavini E (2019) Polyhydroxyalkanoates and crotonic acid from anaerobically digested sewage sludge. ACS Sustain Chem Eng 7(12):10266–10273. https://doi.org/10.1021/acssuschemeng.8b06615
Sivakanthan S, Rajendran S, Gamage A, Madhujith T, Mani S (2020) Antioxidant and antimicrobial applications of biopolymers: a review. Food Res Int 136:109327. https://doi.org/10.1016/j.foodres.2020.109327
Smoker JA, Barnum SR (1988) Rapid small-scale DNA isolation from filamentous cyanobacteria. FEMS Microbiol Lett 56(1):119–122. https://doi.org/10.1111/j.1574-6968.1988.tb03161.x
Tan D, Wang Y, Tong Y, Chen GQ (2021) Grand challenges for industrializing polyhydroxyalkanoates (PHAs). Trends Biotechnol 39(9):953–963. https://doi.org/10.1016/j.tibtech.2020.11.010
Thilakan B, Chakraborty K, Chakraborty R (2016) Antimicrobial properties of cultivable bacteria associated with seaweeds in the Gulf of Mannar on the southeast coast of India. Can J Microbiol 62(8):668–681. https://doi.org/10.1139/cjm-2015-0769
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173(2):697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
Zakaria MR, Ariffin H, Johar NAM, Abd-Aziz S, Nishida H, Shirai Y, Hassan MA (2010) Biosynthesis and characterization of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) copolymer from wild-type Comamonas sp. EB172. Polym Degrad Stabil 95(8):1382–1386. https://doi.org/10.1016/j.polymdegradstab.2010.01.020
Author Information
Rhizosphere Biology Laboratory, Department of Microbiology, Bharathidasan University, Trichy, India