Development of an efficient transformation system in eggplant (Solanum melongena L.) using the phosphomannose isomerase (PMI) gene as the selectable marker
*Article not assigned to an issue yet
Gande Kranthikumar, Marapaka Vasudha, Jogam Phanikanth, Peddaboina Venkataiah
Research Articles | Published: 16 March, 2026
First Page: 0
Last Page: 0
Views: 58
Keywords: Cotyledon, Eggplant, PMI gene, β-glucuronidase, Chlorophenol red assay
Abstract
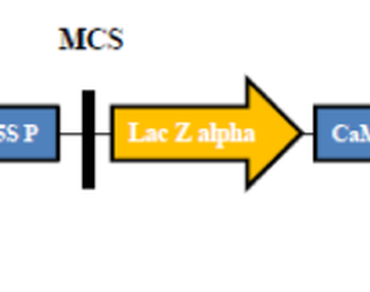
A highly efficient Agrobacterium-mediated genetic transformation procedure has been demonstrated in eggplant (Solanum melongena L.) using the phosphomannose isomerase (PMI/ManA) gene, which was employed as a selectable marker using mannose as a source of carbohydrate. The pre-cultured cotyledons were dipped for 10 min in the suspension of Agrobacterium tumefaciens strain LBA4404 with the PMI gene construct, then co-cultivated for two days. The co-cultivated explants were shifted to the selection medium containing mannose (10 g/l), sucrose (20 g/l), and cefotaxime (500 mg/l) cultured and putatively transgenic shoots were recovered after four to six weeks. The putatively transgenic shoots were rooted on the medium containing mannose (10 g/l), sucrose (20 g/l) and IAA (5.71 µM). The putative transgenic plants were hardened and acclimatized under greenhouse conditions. The T0 transgenic plants showed the stable integration of the PMI gene using PCR analysis. The transgenic plants were validated using histochemical gus assay. The leaf segments of putatively transformed plants were examined with chlorophenol red assay, and the transformed plants expressing the PMI gene changed the red colour to orange or yellow. The transgenic plantlets with the PMI gene exhibited morphologically similar characteristics with the control plants. The present transformation procedure using the PMI gene as a selectable marker gene will be feasible for generating novel germplasm with different candidate genes with genetic transformation and genome editing tools devoid of several antibiotics or herbicides.
References
Alam I, Salimullah M (2021) Genetic Engineering of Eggplant (Solanum melongena L.): Progress, Controversy and Potential. Horticulturae 7:78. https://doi.org/10.3390/horticulturae7040078
Anand A, Bass SH, Wu E, Wang N, McBride KE, Annaluru N, Miller M, Hua M, Jones TJ (2018) An improved ternary vector system for Agrobacterium-mediated rapid maize transformation. Plant Mol Biol 97:187–200. https://doi.org/10.1007/s11103-018-0732-y
Animasaun DA, Adedibu PA, Shkryl Y, Emmanuel FO, Tekutyeva L, Balabanova L (2023) Modern plant biotechnology: an antidote against global food insecurity. Agronomy 13(8):2038. https://doi.org/10.3390/agronomy13082038
Aswath CR, Mo SY, Kim DH, Park SW (2006) Agrobacterium and biolistic transformation of onion using non-antibiotic selection marker phosphomannose isomerase. Plant Cell Rep 25:92–99. https://doi.org/10.1007/s00299-005-0022-4
Aubriot X, Knapp S (2022) A revision of the spiny solanums of Tropical Asia (Solanum, the Leptostemonum Clade, Solanaceae). PhytoKeys 198:1–270. https://doi.org/10.3897/phytokeys.198.79514
Bakshi S, Saha B, Roy NK, Mishra S, Panda SK, Sahoo L (2012) Successful recovery of transgenic cowpea (Vigna unguiculata) using the 6-phosphomannose isomerase gene as the selectable marker. Plant Cell Rep 31:1093–1103. https://doi.org/10.1007/s00299-012-1230-3
Ballester A, Cervera M, Pena L (2008) Evaluation of selection strategies alternative to nptII in genetic transformation of citrus. Plant Cell Rep 27:1005–1015. https://doi.org/10.1007/s00299-008-0523-z
Bello AS, Huda S, Alsafran M, Abu-Dieyeh MH, Chen ZH, Ahmed T (2024) Enhancing eggplant (Solanum melongena L.) yield and water use efficiency through optimized irrigation and nitrogen practices in open field conditions. J Agric Food Res 18:101527. https://doi.org/10.1016/j.jafr.2024.101527
Breyer D, Kopertekh L, Reheul D (2014) Alternatives to antibiotic resistance marker genes for in vitro selection of genetically modified plants – Scientific developments, current use, operational access and biosafety considerations. CRC Crit Rev Plant Sci 33(4):286–330. https://doi.org/10.1080/07352689.2013.870422
Briza J, Pavingerova D, Prikrylova P, Gazdova J, Vlasak J, Niedermeierova H (2008) Use of phosphomannose isomerase-based selection system for Agrobacterium-mediated transformation of tomato and potato. Biol Plant 52:453–461. https://doi.org/10.1007/s10535-008-0090-8
Briza J, Ruzickova N, Niedermeierova H, Dusbabkova J, Vlasak J (2010) Phosphomannose isomerase gene for selection in lettuce (Lactuca sativa L.) transformation. Acta Biochim Pol 57:63–68
Cao B, Huang Z, Chen G, Lei J (2010) Restoring pollen fertility in transgenic male sterile eggplant by Cre/loxp-mediated site-specific recombination system. Genet Mol Biol 33:298–307. https://doi.org/10.1590/S1415-47572010005000043
Degenhardt J, Poppe A, Montag J, Szankowski I (2006) The use of the phosphomannose isomerase/mannose selection system to recover transgenic apple plants. Plant Cell Rep 25:1149–1156. https://doi.org/10.1007/s00299-006-0179-5
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12(1):13–15
Ehsasatvatan M, Jafari M, Darvishzadeh R (2015) Efficient use of the PMI/mannose selection system in Agrobacterium-mediated transformation of tobacco (Nicotiana tabacum). Biologia 70(4):428–437. https://doi.org/10.1515/biolog-2015-0064
FAO (2024) http://www.fao.org/faostat/en/#data/QC. Accessed on 30 Dec 2024
Feeney M, Punja ZK (2003) Tissue culture and Agrobacterium-mediated transformation of hemp (Cannabis sativa L). Vitro Cell Dev Biol Plant 39:578–585. https://doi.org/10.1079/IVP2003454
Gadaleta A, Giancaspro A, Blechl A, Blanco A (2006) Phosphomannose isomerase, PMI, as a selectable marker gene for durum wheat transformation. J Cereal Sci 43:31–37. https://doi.org/10.1016/j.jcs.2005.06.004
Grootboom AW, Mkhonza NL, O’Kennedy MM, Chakauya E, Kunert KJ, Chikwamba RK (2010) Biolistic mediated sorghum (Sorghum bicolor L. Moench) transformation via mannose and bialaphos based selection systems. Int J Bot 6:89–94. https://doi.org/10.3923/ijb.2010.89.94
Gui HP, Li X, Liu YB, Han K, Li XG (2014) The relationship between PMI (manA) gene expression and optimal selection pressure in Indica rice transformation. Plant Cell Rep 33:1081–1090. https://doi.org/10.1007/s00299-014-1596-5
Guo Q, Ma J, Yuan B, Zhou M, Wu Y (2015) High-efficiency Agrobacterium-mediated transformation of Lotus corniculatus L. using phosphomannose isomerase positive selection. Plant Cell Tiss Org Cult 121:413–423. https://doi.org/10.1007/s11240-015-0712-8
Gurel S (2021) Sand-wounding of shoot and petiole explants enhances transformation efficiency in Sugar Beet (Beta vulgaris L.) by Agrobacterium-mediated transformation. Sugar Tech 23(2):415–427. https://doi.org/10.1007/s12355-020-00893-0
He Z, Duan Z, Liang W, Chen F, Yao W, Liang H, Yue C, Sun Z, Chen F, Dai J (2006) Mannose selection system used for cucumber transformation. Plant Cell Rep 25:953–958. https://doi.org/10.1007/s00299-006-0156-z
Hilgert J, Jong MS, Fiser J, Tupa K, Vrbova M, Griga M, Macek T, Ziarovska J (2017) The use of phosphomannose isomerase selection system for Agrobacterium-mediated transformation of tobacco and flax aimed for phytoremediation. J Environ Sci Health Part-B 52(5):338–345. https://doi.org/10.1080/03601234.2017.1283136
Hou J, Chen H, Fang Y, Zhu Y, Han B, Sun C, Fu Y (2021) An Agrobacterium-mediated non-antibiotic selection-based transformation system for rice (Oryza sativa ssp. indica) cultivar 93 – 11 successfully produces TAC1-silenced transgenic plants. Vitro Cell Dev Biol-Plant 57:786–795. https://doi.org/10.1007/s11627-021-10202-3
Hu L, Li H, Qin R, Xu R, Li J, Li L, Wei P, Yang J (2016) Plant phosphomannose isomerase as a selectable marker for rice transformation. Sci Rep 6:25921. https://doi.org/10.1038/srep25921
Hur S-H, Min B-W (2015) Efficient development of transgenic Cabbage with Jasmonic acid carboxyl methyltransferase (JMT) gene based on PMI/Mannose selection system. Plant Breed Biotech 3(3):226–237. https://doi.org/10.9787/PBB.2015.3.3.226
Jefferson RA (1989) The GUS reporter gene system. Nature 342(6251):837–838. https://doi.org/10.1038/342837a0
Khatun M, Borphukan B, Alam I, Keya CA, Khan H, Reddy MK, Salimullah, Md (2022) An improved Agrobacterium-mediated transformation and regeneration protocol for successful genetic engineering and genome editing in eggplant. Scie Horti 293:110716. https://doi.org/10.1016/j.scienta.2021.110716
Kim JY, Jung M, Kim HS, Lee YH, Choi SH, Lim YP, Min BW, Yang SG, Harn CH (2002) A new selection system for pepper regeneration by mannose. J Plant Biotech 4:129–134
Lamblin F, Aime A, Hano C, Roussy I, Domon JM, Van Droogenbroeck B, Laine E (2007) The use of the phosphomannose isomerase gene as alternative selectable marker for Agrobacterium-mediated transformation of flax (Linum usitatissimum). Plant Cell Rep 26:765–772. https://doi.org/10.1007/s00299-006-0280-9
Lin Y, Huang J (2021) Characterization of an algal phosphomannose isomerase gene and its application as a selectable marker for genetic manipulation of tomato. Plant Divers 43:63–70. https://doi.org/10.1016/j.pld.2020.06.001
Min BW, Cho YN, Song MJ, Noh TK, Kim BK, Chae WK, Park YS, Choi YD, Harn CH (2007) Successful genetic transformation of Chinese cabbage using phosphomannose isomerase as a selection marker. Plant Cell Rep 26:337–344. https://doi.org/10.1007/s00299-006-0247-x
Mottaleb KA, Kruseman G, Frija A, Sonder K, Lopez-Ridaura S (2023) Projecting wheat demand in China and India for 2030 and 2050: Implications for food security. Front Nutr 9:1077443. https://doi.org/10.3389/fnut.2022.1077443
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Patil G, Deokar A, Jain PK, Thengane RJ, Srinivasan R (2009) Development of a phosphomannose isomerase-based Agrobacterium-mediated transformation system for chickpea (Cicer arietinum L). Plant Cell Rep 28:1669–1676. https://doi.org/10.1007/s00299-009-0766-3
Penna S, Ganapathi TR (2010) Engineering the plant genome:Prospects of selection systems using non-antibiotic marker genes. GM Crops 1(3):1–9. https://doi.org/10.4161/gmcr.1.3.12383
Prabhavathi V, Rajam MV (2007a) Mannitol-accumulating transgenic eggplants exhibit enhanced resistance to fungal wilts. Plant Sci 173:50–54. https://doi.org/10.1016/j.plantsci.2007.04.004
Prabhavathi V, Rajam MV (2007b) Polyamine accumulation in transgenic eggplant enhances tolerance to multiple abiotic stresses and fungal resistance. Plant Biotech 24:273–282. https://doi.org/10.5511/plantbiotechnology.24.273
Ramesh SA, Kaiser BN, Franks T, Collins G, Sedgley M (2006) Improved methods in Agrobacterium-mediated transformation of almond using positive (mannose/pmi) or negative (kanamycin resistance) selection-based protocols. Plant Cell Rep 25:821–828. https://doi.org/10.1007/s00299-006-0139-0
Reed J, Privalle L, Powell ML, Meghji M, Dawson J, Dunder E, Suttie J, Wenck A, Launis K, Kramer C, Chang YF, Hansen G, Wright M (2001) Phosphomannose isomerase: An efficient selectable marker for plant transformation. Vitro Cell Dev Biol-Plant 37:127–132. https://doi.org/10.1007/s11627-001-0024-z
Rong RJ, Wu PC, Lan JP, Wei HF, Wei J, Chen H, Shi JN, Hao YJ, Liu LJ, Dou SJ, Li LY, Wu L, Liu SQ, Yin CC, Liu GZ (2016) Western blot detection of PMI protein in transgenic rice. J Integr Agric 15:726–734. https://doi.org/10.1016/S2095-3119(15)61053-X
Sadhu S, Jogam P, Thampu RK, Abbagani S, Penn S, Peddaboina V (2020) High efficiency plant regeneration and genetic fidelity of regenerants by SCoT and ISSR markers in chickpea (Cicer arietinum L). Planr Cell Tissue Organ Cult 141:465–477. https://doi.org/10.1007/s11240-020-01804-7
Saini DK, Kaushik P (2019) Visiting eggplant from a biotechnological perspective: A review. Scientia Horti 253:327–340. https://doi.org/10.1016/j.scienta.2019.04.042
Sharma S, Sharma A, Katoch V (2020) Biotechnological interventions in eggplant (Solanum melongena L). J Horti Sci Biotech 95(3):273–285. https://doi.org/10.1080/14620316.2019.1686432
Shi F, Tan M, Mo Q, Chen X, Zhao Y, Guo X, Jiang N (2020) Application of the phosphomannose-isomerase/mannose selection system in the Agrobacterium-mediated transformation of Lonicera hypoglauca Miq. J Plant Biochem Biotech 29(3):528–538. https://doi.org/10.1007/s13562-020-00570-z
Sidorova T, Mikhailov R, Pushin A, Miroshnichenko D, Dolgov S (2017) A non-antibiotic selection strategy uses the phosphomannose-isomerase (PMI) gene and green fluorescent protein (GFP) gene for Agrobacterium-mediated transformation of Prunus domestica L. leaf explants. Plant Cell Tiss Organ Cult 128:197–209. https://doi.org/10.1007/s11240-016-1100-8
Simatele MD, Tantoh HB, Donkor FK (2023) Editorial: Climate change, land, energy and food security: perspectives from Sub-Saharan Africa. Front Sustain Food Syst 7:1164917. https://doi.org/10.3389/fsufs.2023.1164917
Stoykova P, Stoeva-Popova P (2011) PMI (manA) as a nonantibiotic selectable marker gene in plant biotechnology. Plant Cell Tiss Organ Cult 105:141–148. https://doi.org/10.1007/s11240-010-9858-6
Súnico V, Piunti I, Bhattacharjee M, Mezzetti B, Caballero JL, Munoz-Blanco J, Ricci A, Sabbadini S (2024) Overview on current selectable marker systems and novel marker free approaches in fruit tree genetic engineering. Int J Mol Sci 25:11902. https://doi.org/10.3390/ijms252211902
Taher D, Solberg SO, Prohens J, Chou Y, Rakha M, Wu T (2017) World vegetable center eggplant collection: origin, composition, seed dissemination and utilization in breeding. Front Plant Sci 8:1484. https://doi.org/10.3389/fpls.2017.01484
Todd R, Tague BW (2001) Phosphomannose isomerase: a versatile selectable marker for Arabidopsis thaliana germ-line transformation. Plant Mol Biol Rep 19:307–319. https://doi.org/10.1007/BF02772829
Usha S, Sachin S (2025) Biotechnological precision in agriculture and food traceability. In: Gaikwad PS, Choudhary P (eds) Biotechnological Innovations in Food Processing. Springer, Cham. https://doi.org/10.1007/978-3-031-96526-5_13.
Wallbraun M, Sonntag K, Eisenhauer C, Krzcal G, Wang YP (2009) Phosphomannose-isomerase (pmi) gene as a selectable marker for Agrobacterium-mediated transformation of rapeseed. Plant Cell Tiss Organ Cult 99:345–351. https://doi.org/10.1007/s11240-009-9610-2
Wang AS, Evans RA, Altendorf PR, Hanten JA, Doyle MC, Rosichan JL (2000) A mannose selection system for production of fertile transgenic maize plants from protoplasts. Plant Cell Rep 19:654–660. https://doi.org/10.1007/s002999900181
Wang H, Petri C, Burgos L, Alburquerque N (2013) Phoshomannoseisomerase as a selectable marker for transgenic plum (Prunus domestica L). Plant Cell Tissue Org Cult 133:189–197. https://doi.org/10.1007/s11240-012-0259-x
Wang WZ, Yang BP, Feng XY, Cao ZY, Feng CL, Wang JG, Xiong GR, Shen LB, Zeng J, Zhao TT, Zhang SZ (2017) Development and characterization of transgenic sugarcane with insect resistance and herbicide tolerance. Front Plant Sci 8:1535. https://doi.org/10.3389/fpls.2017.01535
Wang Y, Geng L, Gui H, Liu Y, Wang Y, Zhang X, Li X (2015) Soybean PMI genes as a selectable marker for corn and rice transformation. Maize Genet Cooperation Newsl 89:1–3. https://mnl.maizegdb.org/89/pdf/13wang.pdf
Weigel D, Glazebrook J (2006) Transformation of Agrobacterium using freeze-thaw method. Cold Spring Harb Protoc. https://doi.org/10.1101/pdb.prot4666
Wright M, Dawson J, Dunder E, Suttie J, Reed J, Kramer C, Chang Y, Novitzky R, Wang H, Artim-Moore L (2001) Efficient biolistic transformation of maize (Zea mays L.) and wheat (Triticum aestivum L.) using the phosphomannose isomerase gene, pmi, as the selectable marker. Plant Cell Rep 20:429–436. https://doi.org/10.1007/s002990100318
Wu H, Acanda Y, Canton M, Zale J (2019) Efficient Biolistic Transformation of Immature Citrus Rootstocks Using Phosphomannose-isomerase Selection. Plants 8(10):390. https://doi.org/10.3390/plants8100390
Wu H, Canton M, Mahmoud LM, Weber KR, Michalczyk GZ, Dutt M, Zale JM (2022) Identification and characterization of two putative Citrus Phosphomannose Isomerase (CsPMI) genes as selectable markers for mature citrus transformation. Horticulturae 8:204. https://doi.org/10.3390/horticulturae8030204
Yesmin S, Hoque MI, Sarker RH (2021) Enhanced regeneration through ex vitro rooting and Agrobacterium-mediated genetic transformation of eggplant (Solanum melongena L). Plant Tissue Cult Biotech 31(1):97–108. https://doi.org/10.3329/ptcb.v31i1.54115
Author Information
Department of Microbiology, Kakatiya University, Vidyaranyapuri, Warangal, India