Soil properties determine secondary metabolite composition in Brassica juncea (Indian mustard)
*Article not assigned to an issue yet
Research Articles | Published: 05 March, 2026
First Page: 0
Last Page: 0
Views: 77
Keywords: n Brassica juncean , Indian mustard, Secondary metabolites, Phytochemical analysis, GC‒MS, Soil composition
Abstract
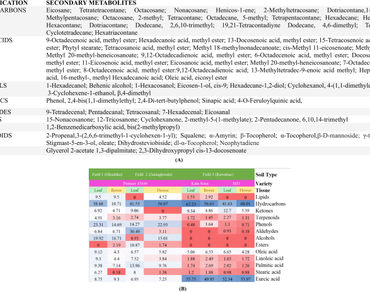
Indian mustard is widely used in culinary applications because of its nutraceutical properties as an anti-inflammatory, anti-cardiovascular, anti-carcinogenic, anti-microbial, anti-diarrheal, & anti-ulcer agent. It is also a valuable cash crop for edible/industrial oil production. An increase in the productivity of oil seed crops, including the widely used Brassica juncea (Indian mustard), is a priority, as the import of edible oils is steadily increasing to meet the ever-growing domestic demand. The present study investigated the effects of soil type on the secondary metabolite composition and concentration in mustard leaf and flower tissues. Different mustard varieties (Kala Sona and MJ-1 varieties) grown on the same kind of soil and a single variety of mustard (Pioneer 45S46 variety) grown under two different edaphic conditions presented significant variations in secondary metabolite composition as well as concentrations in leaf and flower tissues, as determined by GC‒MS analysis. Interactions between soil and plants, facilitated by secondary metabolites, can affect plant–insect relationships, which in turn can impact the productivity of mustard.
References
Bustamante MÁ, Michelozzi M, Barra Caracciolo A, Grenni P, Verbokkem J, Geerdink P, Nogués I (2020) Effects of soil fertilization on terpenoids and other carbon-based secondary metabolites in Rosmarinus officinalis plants: a comparative study. Plants 9(7):830. https://doi.org/10.3390/plants9070830
Chaouqi S, Moratalla-López N, Alonso GL, Lorenzo C, Zouahri A, Asserar N, Haidar EM, Guedira T (2023) Effect of soil composition on secondary metabolites of Moroccan saffron (Crocus sativus L.). Plants 12(4):711. https://doi.org/10.3390/plants12040711
Dixon RA, Dickinson AJ (2024) A century of studying plant secondary metabolism—from “what?” to “where, how, and why?” Plant Physiol 195(1):48–66. https://doi.org/10.1093/plphys/kiad596
Galanty A, Grudzińska M, Paździora W, Paśko P (2023) Erucic acid—both sides of the story: a concise review on its beneficial and toxic properties. Molecules 28(4):1924. https://doi.org/10.3390/molecules28041924
Gfeller V, Waelchli J, Pfister S, Deslandes-Hérold G, Mascher F, Glauser G, Erb M (2023) Plant secondary metabolite-dependent plant-soil feedbacks can improve crop yield in the field. Elife 12:e84988. https://doi.org/10.7554/elife.84988
González Mera IF, González Falconí DE, Morera Córdova V (2019) Secondary metabolites in plants: main classes, phytochemical analysis, and pharmacological activities. Rev Bionatura 4(4):1000–1009. https://doi.org/10.21931/RB/2019.04.04
Harbort CJ, Hashimoto M, Inoue H, Niu Y, Guan R, Rombolà AD, Schulze-Lefert P (2020) Root-secreted coumarins and the microbiota interact to improve iron nutrition in Arabidopsis. Cell Host Microbe 28(6):825–837. https://doi.org/10.1016/j.chom.2020.09.006
Hu L, Robert CA, Cadot S, Zhang XI, Ye M, Li B, Erb M (2018) Root exudate metabolites drive plant-soil feedbacks on growth and defense by shaping the rhizosphere microbiota. Nat Commun 9(1):2738. https://doi.org/10.1038/s41467-018-05122-7
Hu L, Wu Z, Robert CAM, Ouyang X, Züst T, Mestrot A, Xu J, Erb M (2021) Soil chemistry determines whether defensive plant secondary metabolites promote or suppress herbivore growth. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.2105329118
Huang AC, Jiang T, Liu YX, Bai YC, Reed J, Qu B, Osbourn A (2019) A specialized metabolic network selectively modulates Arabidopsis root microbiota. Science 364(6440):eaau6389. https://doi.org/10.1126/science.aau6389
Indriati S, Hidayat MY, Riskayanti R, Amaliah N, Latief M, Sjafruddin R, Attahmid NF (2021) GC–MS and antioxidant capacity analysis in propanol extract of Carthamus tinctorius L. INTEK: Jurnal Penelitian 8:67. https://doi.org/10.31963/intek.v8i1.2801
Jaafar HZ, Ibrahim MH, Fakri NFM (2012) Impact of soil field water capacity on secondary metabolites, phenylalanine ammonia-lyase (PAL), maliondialdehyde (MDA) and photosynthetic responses of Malaysian Kacip Fatimah (Labisia pumila Benth). Molecules 17(6):7305–7322. https://doi.org/10.3390/molecules17067305
Kabera JN, Semana E, Mussa AR, He X (2014) Plant secondary metabolites: biosynthesis, classification, function, and pharmacological properties. J Pharm Pharmacol 2(7):377–392
Khalid KA, Darwesh OM, Ahmed AM (2021) Peel essential oils of citrus types and their antimicrobial activities in response to various growth locations. J Essent Oil-Bear Plants 24(3):480–499. https://doi.org/10.1080/0972060X.2021.1926303
Kopaczyk JM, Warguła J, Jelonek T (2020) The variability of terpenes in conifers under developmental and environmental stimuli. Environ Exp Bot 180:104197. https://doi.org/10.1016/j.envexpbot.2020.104197
Koprivova A, Schuck S, Jacoby RP, Klinkhammer I, Welter B, Leson L, Kopriva S (2019) Root-specific camalexin biosynthesis controls the plant growth-promoting effects of multiple bacterial strains. Proc Natl Acad Sci U S A 116(31):15735–15744. https://doi.org/10.1073/pnas.1818604116
Kumar S (2017) Plant secondary metabolites (PSMs) of Brassicaceae and their role in plant defense against insect herbivores–a review. J Appl Nat Sci 9(1):508. https://doi.org/10.31018/jans.v9i1.1245
Kumari A, Sharma S, Sharma M (2018) Effect of physicochemical properties of soil on secondary metabolites of Calotropis gigantea (L) collected from Shivalik hills of Himachal Pradesh, India. Biol Forum 10(1):146–157
Manzano-Gómez LA, Guzmán-Albores JM, Rincón-Rosales R, Winkler R, Rincón-Molina CI, Castañón-González JH, Ruíz-Valdiviezo VM (2021) Evaluation of metabolomic profile and growth of Moringa oleifera L. cultivated with vermicompost under different soil types. Agronomy 11(10):2061. https://doi.org/10.3390/agronomy11102061
Mehalaine S, Chenchouni H (2020) Plants of the same place do not have the same metabolic pace: soil properties affect differently essential oil yields of plants growing wild in semiarid Mediterranean lands. Arab J Geosci 13(23):1263. https://doi.org/10.1007/s12517-020-06318-9
Mumivand H, Babalar M, Hadian J, Fakhr-Tabatabaei M (2011) Plant growth and essential oil content and composition of Satureja hortensis L. cv. Saturn in response to calcium carbonate and nitrogen application rates. J Med Plants Res 5(10):1859–1866
Ormeño E, Fernandez C (2012) Effect of soil nutrient on production and diversity of volatile terpenoids from plants. Curr Bioact Comp 8(1):71–79
Palma-López J, Sánchez-Rodríguez AR, del Campillo MC, León-Gutiérrez JM, Ramírez-Pérez P (2024) Influence of soil properties on grape and must quality in the Montilla-Moriles protected designation of origin (southern Spain). CATENA 241:108041. https://doi.org/10.1016/j.catena.2024.108041
Parikh SJ, James BR (2012) Soil: the foundation of agriculture. Nat Edu Know 3(10):2
Author Information
Department of Life Science, Central University of Karnataka, Kalaburagi, India