The effect of sonication times on bioactive compounds, antioxidant activity and phenolic constituents of sumac (Rhus coriaria L.) fruits extracted by ultrasound extraction system
*Article not assigned to an issue yet
Mohamed Ahmed Isam A., AlJuhaimi Fahad, Özcan Mehmet Musa, Uslu Nurhan, Karrar Emad
Research Articles | Published: 23 March, 2026
First Page: 0
Last Page: 0
Views: 435
Keywords: Sumac fruit, Sonication, Bioactive compounds, Antioxidant activity, Phenolic compounds, HPLC
Abstract
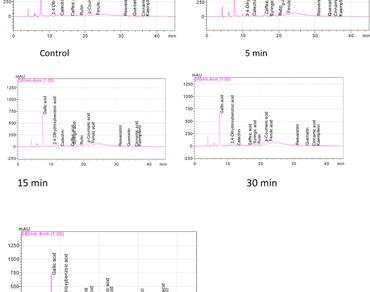
In current investigation, the bioactive compounds, antioxidant capacities (DPPH and FRAP) and phenolic constituents of sumac fruits sonicated at different times (5,15, 30 and 45 min.) were determined. Total phenolic and flavonoid contents of sumac fruits sonicated at different times changed between 1275.24 (45 min) and 1481.59 mg GAE/100 g (15 min) to 1295.71 (5 min) and 1427.14 mg/100 g (15 min), respectively. While the total phenol content of sumac fruits increased until the 15th minute of sonication, it decreased in the following periods. However, it was observed that the total flavonoid contents of sumacs increased with the increase of sonication time (except 5 min.). The highest total phenol and flavonoid contents were assigned at the 15th minute of sonication. Antioxidant activities (DPPH and FRAP) of sumac fruits sonicated at different times changed between 16.37 (5 min) and 16.49 mmol/kg (45 min.) to 120.77 (5 min.) and 124.35 mg/g (15 min.), respectively. Results on phenolic compounds exhibited some differences depending on different sonication times. Except for cinnamic acid and kaempferol, the phenolic component amounts of the control and sumacs sonicated at different times were quite high. The phenolic components of sumac fruit increased with the sonication time. Gallic acid and catechin amounts of sumach fruits sonicated at different sonication times were detected to be between 1258.82 (45 min.) and 2012.80 mg/100 g (15 min.) to 238.76 (30 min.) and 629.80 mg/100 g (45 min.), respectively. In general, the highest quantities of phenolic compounds were observed at the 15th and 30th minutes of sonication.
References
Abdel-Mawgoud M, Khedr FG, Mohammed EI (2019) Phenolic compounds, antioxidant and antibacterial activities of Rhus flexicaulis Baker. J J Biol Sci 12:17–21.ISSN 1995–6673
Ali-Shtayeh MS, Al-Assali AA, Jamous RM (2013) Antimicrobial activity of Palestinian medicinal plants against acne-inducing bacteria. Afr J Microbiol Res 7:2560–2573. https://doi.org/10.5897/AJMR12.1875
Al-Muwaly KY, Khawola A, Flayeh A (2013) Antioxidant and free radical scavenging effects of Iraqi sumac (Rhus coriaria L.). Baghdad Sci J 10:921–933. https://doi.org/10.21123/bsj.2013.10.3.921-933
Alsamri H, Athamneh K, Pintus G, Eid AH, Iratni R (2021) Pharmacological and antioxidant activities of Rhus coriaria L. (Sumac). Antioxidants 10:73. https://doi.org/10.3390/antiox10010073
Ardalani H, Moghadam MH, Rahimi R, Soltani J, Mozayanimonfared A, Moradi M, Azizi A (2016) Sumac as a novel adjunctive treatment in hypertension: a randomized, double-blind, placebo-controlled clinical trial. RSC Adv 6:11507
Bashash M, Zamindar N, Bolandi M (2014) Evaluation of antioxidant activities of Iranian sumac (R. coriaria L.) fruit and spice extracts with different solvents. J Food Measur Charact 8:213–217. https://doi.org/10.1007/s11694-014-9182-7
Bashash M, Bolandi M, Zamindar N (2012) Phenolic Content of Selected Sumac Fruits from Iran, Extracted With Different Solvents. J Chem Health Risks 2(4): 17–20. ISSN:2251–6719
Bloshenko EK, Letchamo W (1995) Characterization of natural distribution and some biological traits of Sumach (Rhus coriaria) in central Asia. In international symposium on medicinal and aromatic plants 426:113–122
Bozkurt H (2006) Investigation of the effect of sumac extract and BHT addition on the quality of sucuk (Turkish dry-fermented sausage). J Sci Food Agric 86:849–856. https://doi.org/10.1002/jsfa.2431
Bursal E, Köksal E (2011) Evaluation of reducing power and radical scavenging activities of water and ethanol extracts from sumac (Rhus coriaria L.). Food Res Int 44(7):2217–2221. https://doi.org/10.1016/j.foodres.2010.11.001
Büyüktuncel E (2012) Gelişmiş Ekstraksiyon Teknikleri I. Hacettepe Univ Ecz Fak Derg 32(2):209–242
Çalışkan R, Sarı SP, Büyükkılıç Altınbaşak B, Dinç HÖ, Balekoğlu A, Issa G, Yüksel Mayda P (2022) Bioactive components and antioxidant and antimicrobial activities of Rhus coriaria, a sumac species found in Turkey. Bezmialem Sci 10(6):796–804. https://doi.org/10.14235/bas.galenos.2022.84803
Candan F, Sökmen A (2004) Effect of Rhus coriaria L. (Anacardiaceae) on lipid peroxidation and free radical scavenging activity. Phytother Res 18(1):84–86. https://doi.org/10.1002/ptr.1228
Carrera C, Ruiz-Rodríguez A, Palma M, Barroso CG (2012) Ultrasound assisted extraction of phenolic compounds from grapes. Anal Chim Acta 732:100–104. https://doi.org/10.1016/j.aca.2011.11.032
Chakraborty A, Ferk F, Simić T, Brantner A, Dušinská M, Kundi M, Hoelzl C, Nersesyan A, Knasmüller S (2009) DNA-protective effects of sumac (Rhus coriaria L.), a common spice: results of human and animal studies. Mutat Res-Fund Mol Mech Mutagen 661:10–17. https://doi.org/10.1016/j.mrfmmm.2008.10.009
Corrado G, El-Nakhel C, Graziani G, Pannico A, Zarrelli A, Giannini P, Ritieni A, De Pascale S, Kyriacou MC, Rouphael Y (2021) Productive and morphometric traits, mineral composition and secondary metabolome components of Borage and Purslane as underutilized species for microgreens production. Horticulturae 7:211. https://doi.org/10.3390/horticulturae7080211
Doğan M, Akgül A (2005) Characteristics and fatty acid composition of Rhus coriaria cultivars from southeast Turkey. Chem Nat Compd 41:724–725. https://doi.org/10.1007/s10600-006-0021-x
Faller ALK, Fialho E (2009) The antioxidant capacity and polyphenol content of organic and conventional retail vegetables after domestic cooking. Food Res Int 42(1):210–215. https://doi.org/10.1016/j.foodres.2008.10.009
Fazeli MR, Amin G, Attari MMA, Ashtiani H, Jamalifar H, Samadi N (2007) Antimicrobial activities of Iranian sumac and avishan-e shirazi (Zataria multiflora) against some food-borne bacteria. Food Control 18:646–649. https://doi.org/10.1016/j.foodcont.2006.03.002
Fereidoonfar H, Salehi-Arjmand H, Khadivi A, Akramian M, Safdari L (2019) Chemical variation and antioxidant capacity of sumac (Rhus coriaria L.). Ind Crops Prod 139:111518. https://doi.org/10.1016/j.indcrop.2019.111518
Güner A (2012) Türkiye Bitkiler Listesi Damarlı Bitkileri, Nezahat Gökyiğit Botanik Bahçesi Yayınları, İstanbul.
Hogan S, Zhang L, Li J, Zoecklein B, Zhou K (2009) Antioxidant properties and bioactive components of Norton (Vitis aestivalis) and Cabernet Franc (Vitis vinifera) wine grapes. LWT - Food Sci Technol 42:1269–1274. https://doi.org/10.1016/j.lwt.2009.02.006
Itidel C, Chokri M, Mohamed B, Yosr Z (2013) Antioxidant activity, total phenolic and flavonoid content variation among Tunisian natural populations of Rhus tripartita (Ucria) Grande and Rhus pentaphylla Desf. Ind Crops Prod 51:171–177. https://doi.org/10.1016/j.indcrop.2013.09.002
Karadaş Ö, Yılmaz İ, Geçgel Ü (2020) Properiıes of sumac plant and its importance in nutrition. Int J Innov Approaches Agric Res 4(3):377–383. https://doi.org/10.29329/ijiaar.2020.274.10
Karaduman N (2022) Sumak (Rhus coriaria) Bitkisinin Beslenme ve Fitoterapide Kullanımı Üzerine Araştırmalar. Bezmialem Vakıf Üniversitesi Sağlık Bilimleri Enstitüsü Farmakognozi ve Doğal Ürünler Kimyası Anabilim Dalı Farmakognozi ve Doğal Ürünler Kimyası Tezli Yüksek Lisans Programı. Yüksek Lisans Tezi.
Kchaou W, Abbes F, Blecker C, Attia H, Besbes S (2013) Effects of extraction solvents on phenolic contents and antioxidant activities of Tunisian date varieties (Phoenix dactylifera L.). Ind Crops Prod 45:262–269. https://doi.org/10.1016/j.indcrop.2012.12.028
Kosar M, Bozan B, Temelli F, Baser KHC (2007) Antioxidant activity and phenolic composition of sumac (Rhus coriaria L.) extracts. Food Chem 103(3):952–959. https://doi.org/10.1016/j.foodchem.2006.09.049
Kossah R, Nsabimana C, Zhao J, Chen H, Tian F, Zhang H, Chen W (2009) Comparative study on the chemical composition of Syrian sumac (Rhus coriaria L.) and Chinese sumac (Rhus typhina L.) fruits. Pak J Nutr 8:1570–1574. https://doi.org/10.3923/pjn.2009.1570.1574
Lavilla I, Bendicho C (2017) Fundamentals of ultrasound-assisted extraction. Water Extr Bioact Comp from Plants to Drug Dev 2017:291–316. https://doi.org/10.1016/B978-0-12-809380-1.00011-5
Lee SK, Mbwambo ZH, Chung HS, Luyengi L, Games EJC, Mehta RG (1998) Evaluation of the antioxidant potential of natural products. Comb Chem High Throughput Screen 1:35–46
Ma YQ, Ye XQ, Fang ZX, Chen JC, Xu GH, Liu DH (2008) Phenolic compounds and antioxidant activity of extracts from ultrasonic treatment of Satsuma mandarin (Citrus unshiu Marc.) peels. J Agric Food Chem 56:5682–5690. https://doi.org/10.1021/jf072474o
Martins S, Aguilar CN, la De Garza I, Mussatto S, Texeira JA (2010) Kinetic study of nordihydroguaiaretic acid recovery from Larrea tridentata by microwave-assisted extraction. J Chem Technol Biotechnol 85:1142–1147. https://doi.org/10.1002/jctb.2412
Mudenuti NVR, de Camargoa AC, de Alencar SM, Danielski R, Shahidi F, Madeira TB, Hirooka EY, Spinosa WA, Grossmann MVE (2021) Phenolics and alkaloids of raw cocoa nibs and husk: the role of soluble and insoluble-bound antioxidants. Food Biosci 42:101085. https://doi.org/10.1016/j.fbio.2021.101085
Nasar-Abbas SM, Halkman AK (2003) Antimicrobial effect of water extract of sumac (Rhus coriaria L.) on the growth of some food borne bacteria including pathogens. Int J Food Microbiol 97:63–69. https://doi.org/10.1016/j.ijfoodmicro.2004.04.009
Özaydın F, Vardin H (2022) Effects of antioxidant and physicochemical properties on antimicrobial activity of sumac (Rhus coriaria L.) plant spices which are collected from the southeastern anatolia region of Turkey. Harran Tarım Ve Gıda Bilimleri Dergisi 26(3):327–335. https://doi.org/10.29050/harranziraat.1136348
Özcan M (2003a) Effect of sumach (Rhus coriaria L.) extracts on the oxidative stability of peanut oil. J Med Food 6:63–66. https://doi.org/10.1089/109662003765184769
Özcan M (2003b) Antioxidant activities of rosemary, sage, and sumac extracts and their combinations on stability of natural peanut oil. J Med Food 6(3):267–270. https://doi.org/10.1089/10966200360716698
Özcan M (2004) Mineral contents of some plants used as condiments in Turkey. Food Chem 84(3):437–440. https://doi.org/10.1016/S0308-8146(03)00263-2
Özcan M, Haciseferogullari H (2004) A Condiment [Sumac (Rhus coriaria L.) Fruits]: Some Physicochemical Properties. Bulgarian J Plant Physiol 30(3–4): 74–84.
Perrone A, Yousefi S, Basile B, Corrado G, Giovino A, Salami SA, Papini A, Martinelli F (2022) Phytochemical, antioxidant, anti-microbial, and pharmaceutical properties of sumac (Rhus coriaria L.) and its genetic diversity. Hort 8:1168. https://doi.org/10.3390/horticulturae8121168
Rad AH, Khaleghi M, Javadi M (2020) Sumac in food industry: a changing outlook for consumer and producer. J Food Technol Nutr Sci 2(1):1–3
Raodah M, Al-Ali Alia ZH, Faleeha HH (2014) The antioxidant and antimicrobial of syrian sumac (Rhus coriaria) fruit extracts. J Nat Sci Res 4:36–40
Rayne S, Mazza G (2007) Biological activities of extracts from sumac (Rhus spp.): a review. Plant Foods Hum Nutr 62(4):165–175. https://doi.org/10.1007/s11130-007-0058-4
Sakhr K, Khatib SE (2020) Physiochemical properties and medicinal, nutritional and industrial applications of Lebanese Sumac (Syrian Sumac-Rhus coriaria): a review. Heliyon 6(1):e03207. https://doi.org/10.1016/j.heliyon.2020.e03207
Seruga JL, Novak IM (2007) Flavonols, phenolic acid and antioxidant activity of some red fruit. J Food Technol 51:369–378
Setorki M, Rafieian M, Heidarian E, Ghatreh K, Shahinfard N, Ansari R, Forouzandeh Z (2012) Effect of Rhus coriaria consumption with high cholesterol food on some atherosclerosis risk factors in rabbit. J Babol Univ Med Sci 14:38–45
Sezik E, Tabata M, Yesilada E (1991) Traditional medicine in Turkey. 1. Folk medicine in northeast Anatolia. J Ethnopharmacol 35:191–196. https://doi.org/10.1016/0378-8741(91)90072-l
Sultana B, Anwar F, Ashraf M (2009) Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules 14:2167–2180. https://doi.org/10.3390/molecules14062167
Tehrani M, Hesarinejad MA, Razavi SMA, Mohammadian R, Poorkian S (2017) Comparing physicochemical properties and antioxidant potential of sumac from Iran and Turkey. MOJ Food Process Technol 5: 1–7. https://doi.org/10.15406/mojfpt.2017.05.00125
Tohma H, Altay A, Köksal E, Gören AC, Gülçin İ (2019) Measurement of anticancer, antidiabetic and anticholinergic properties of sumac (Rhus coriaria): analysis of its phenolic compounds by LC–MS/MS. J Food Measur Character 13:1607–1619. https://doi.org/10.1007/s11694-019-00077-9
Tsao R, Deng Z (2004) Separation procedures for naturally occurring antioxidant phytochemicals. J Chromatogr B Anal Technol Biomed Life Sci 812(1–2):85–99. https://doi.org/10.1016/j.jchromb.2004.09.028
Ünder D, Saltan FZ (2019) Sumac and its important biological effects. Çukurova J Agric Food Sci 34(1):51–60
Wang L, Weller CL (2006) Recent advances in extraction of nutraceuticals from plants. Trends Food Sci Technol 17:300–312. https://doi.org/10.1016/j.tifs.2005.12.004
Wang L, Xu ML, Guo LJ, Liu J, Wang LS (2015) Antioxidant activities and phenolic compounds of various extracts of Rhus typhina fruits and leaves. Adv J Food Sci Technol 7(4):223–229. https://doi.org/10.19026/ajfst.7.1299
Yoo KM, Lee KW, Park JB, Lee HJ, Hwang IK (2004) Variation in major antioxidants and total antioxidant activity of Yuzu (Citrusjunos SiebexTanaka) during maturation and between cultivars. J Agric Food Chem 52:5907–5913. https://doi.org/10.1021/jf0498158
Zafar MM, Manan A, Razzaq A, Zulfqar M, Saeed A, Kashif M, Khan AI, Sarfraz Z, Mo H, Iqbal MS et al (2021) Exploiting agronomic and biochemical traits to develop heat resilient cotton cultivars under climate change scenarios. Agron 11:1885. https://doi.org/10.3390/agronomy11091885
Author Information
Department of Food Science & Nutrition, College of Food and Agricultural Sciences, King Saud University, Riyadh, Saudi Arabia