TDZ and zeatin induced in vitro shooting for efficient vegetative propagation in Trillium govanianum, an endangered alpine medicinal herb
Chandola Vaishali, Chandra Sudeep, Nautiyal A. R., Kumar Rajeev Ranjan, Purohit V. K.
Research Articles | Published: 23 October, 2023
First Page: 2448
Last Page: 2456
Views: 3454
Keywords: In vitro propagation, Multiple shoot formation, n Trillium govanianumn , TDZ, Zeatin
Abstract
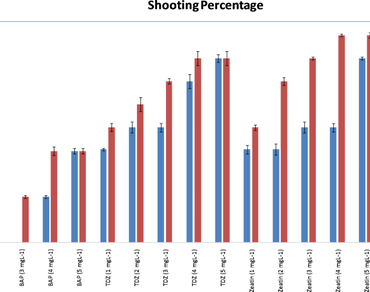
Trillium govanianum, locally known as “Nagchattri”, is an endangered alpine herb and a potent source of plant steroids. Its unabated exploitation and limited seed production are the major threats to its sustainability and calls for methods of conservation. The present study thus focuses on vegetative propagation in T. govanianum and reports effective and repeatable in vitro propagation protocol for multiple shoot proliferation and plantlet formation in it. The study involves the use of biotechnological tools and exogenously supplied plant growth regulators to overcome dormancy in in vitro cultured apical buds of T. govanianum resulting in multiple shoot proliferation. The study also throws light on propagule harvesting stage and chamber conditions for successful proliferation of T. govanianum. Apical buds from dormant period were found more responsive than apical buds from emergence period. Onset of shoot initiation took a minimum of 20 d to a maximum of 54 d whereas complete shoot formation took a minimum of 43 d to a maximum of 96 d. Thidiazuron (TDZ) and Zeatin (Zea) were found to be effective at 4 and 5 mgL-1. Maximum shooting percentage (75%) was observed in the bud explants from dormant period. Maximum shoot formation (3 ± 1) and maximum shoot length (1.8 ± 0.02 cm) was observed in MS media with Zeatin (4 and 5 mgL-1) in dormant buds. Maximum root proliferation (88.9 ± 0.6%) was observed in IAA + TDZ (3 + 1 mgL-1) and took an average of 65 ± 5 d. The results of this study can be employed for producing T. govanianum plants and its efficient in vitro culture and conservation.
References
Alatar A (2015) Thidiazuron induced efficient in vitro multiplication and ex vitro conservation of Rauvolfia serpentina - A potent antihypertensive drug producing plant. Biotechnol Biotechnol Equip 29:1–9. https://doi.org/10.1080/13102818.2015.1017535
Baskin C, Baskin J (2014) Seeds: Ecology, Biogeography, and, evolution of Dormancy and Germination, 2nd edn. Academic Press
Chandola V, Chandra S, Kumar RR et al (2022a) Reproductive behavior of an endangered Himalayan medicinal herb Trillium govanianum Wall. ex D. Don. Vegetos. https://doi.org/10.1007/s42535-022-00548-3
Chandola V, Chandra S, Nautiyal AR et al (2022b) Antioxidant potential and impact of different extraction solvents on the free, esterified and insoluble-bound phenolics, flavonoid and tannin content of Trillium govanianum Wall ex D. Don, a rare Himalayan herb. Vegetos. 35, 953–960 (2022). https://doi.org/10.1007/s42535-022-00365-8
Chandola V, Nautiyal AR, Chandra S, Ranjan R (2019) A review on prioritizing conservation of himalayan medicinal plant species: case of Trillium govanianum (Wall. Ex D. Don) kunth. J Med Plants Stud 7(5):23–27
Chauhan HK, Bisht AK (2020) Trillium govanianum, Himalayan Trillium. The IUCN Red List of Threatened Species 2020 eT175804005A176257695. https://doi.org/10.2305/IUCN.UK.2020-3.RLTS.T175804005A176257695.en
Chauhan HK, Bisht AK, Bhatt ID, Bhatt A (2020) Protocol for vegetative propagation of Trillium govanianum Wall ex D. Don. Journal of Applied Research on Medicinal and Aromatic Plants. 16; https://doi.org/10.1016/j.jarmap.2019.100233
Chauhan NS (1999) Medicinal and aromatic plants of Himachal Pradesh. Indus Publishing, New Delhi, India
Chen X, Xiaodi H, Goa HY, Haiyan W (2003) Study on tissue culture of Rhododendron Simsii planch. J Jiangsu Forestry Sci Technol 30:10–12
Chen Y, Li F (2005) Micropropagation and callus culture of Saussurea laniceps, an alpine medicinal plant. For Stud China 7:16–19. https://doi.org/10.1007/s11632-005-0051-7
Dello Ioio R, Scaglia Linhares F, Scacchi E, Casamitjana-Martinez E, Heidstra R, Costantino P (2007) Cytokinins determine Arabidopsis root-meristem size by controlling cell differentiation. Curr Biol 17:678–682. https://doi.org/10.1016/j.cub.2007.02.047
Gagliardo R, Labarthe M, Zaic M, Cruse-Sanders J, Determann RO (2012) In vitro propagation of Trillium species with notes on root formation, cleaning protocols, and media formulations. Native Plants Journal 13(1):56–63. https://www.muse.jhu.edu/article/470880
Hsia CN, Korban SS (1997) The influence of cytokinins and ionic strength of Anderson’s medium on shoot establishment and proliferation of evergreen azalea. Ephytica 93:11–17
Huetteman C, Preece J (1993) Thidiazuron: a potent cytokinin for woody plant tissue culture. Planr Cell Tissue Organ Cult 33:105–119. https://doi.org/10.1007/BF01983223
Kondo T, Mikubo M, Yamada K, Walck JL, Hidayati SN (2011) Seed dormancy in Trillium camschatcense (Melanthiaceae) and the possible roles of light and temperature requirements for seed germination in forests. Am J Bot 98:215–226. https://doi.org/10.3732/ajb.1000151
Mahmood A, Mahmood A, Malik RN (2012) Indigenous knowledge of medicinal plants from Leepa valley, Azad Jammu and Kashmir. Pakistan J Ethnopharmacology 143(1):338–346
Morrish FV, Vasil V, Vasil IK (1987) Developmental morphogenesis and genetic manipulation in tissue and cell cultures of Gramineae. Adv Genet 24:431–499
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–479
Patial V, Sharma M, Bhattacharya A (2016) Potential of thidiazuron in improved micropropagation of Picrorhiza kurroa– an endangered medicinal herb of alpine Himalaya. Plant Biosystems - An International Journal Dealing with all Aspects of Plant Biology 151:1–8. https://doi.org/10.1080/11263504.2016.1219417
Pence VC, Soukup VG (1993) Factors affecting the initiation of mini-rhizomes from Trillium erectum and T. grandiflorum tissues in vitro. Plant Cell Tissue and Organ Culture 35:229–235. https://doi.org/10.1007/BF00037275
Petrić M, Subotic A, Jevremovic S, Trifunović-Momčilov M (2011) Somatic embryogenesis and bulblet regeneration in snakehead fritillary (Fritillaria meleagris L). Afr J Biotechnol 10:16181–16188. https://doi.org/10.5897/AJB09.807
Rahimi S, Naderi R, Ghaemaghami SA, Kalatejari S, Farham B (2013) Study on effects of different plant growth regulators types in shoot regeneration and node formation of Sutsuki Azalea (Rhododendron indicum): a commercially important Bonsai. Procedia Eng 59:240–246
Rahman S, Ur., Adhikari A, Ismail M, Shah MR, Khurram M, Shahid M et al (2016) Beneficial Effects of Trillium govanianum Rhizomes in Pain and inflammation. Molecules 21:1095. https://doi.org/10.3390/molecules21081095
Rahman SU, Ismail M, Khurram M, Haq IU (2015) Pharmacognostic and ethnomedicinal studies on Trillium govanianum. Pak J Bot 47(si):187–192
Rani S, Rana J, Rana P (2013) Ethnomedicinal plants of Chamba district, Himachal Pradesh, India. J Med Plants Res 7(42):3147–3157
Rashid K, Rashid S, Ganie AH et al (2023) Reproductive biology of Trillium govanianum, an endangered plant species endemic to the Himalaya: implications for conservation, Botany Letters. https://doi.org/10.1080/23818107.2023.2176355
Rashid K, Rashid S, Ganie AH, Nawchoo IA, Khuroo AA (2021) Meiotic studies, pollen fertility and seed set of Trillium govanianum, an endangered endemic plant species of the Himalaya. Cytologia 86(3):245–249. https://doi.org/10.1508/cytologia.86.245
Sharma P, Samant S (2014) Diversity, distribution and indigenous uses of medicinal plants in parbati valley of Kullu district in Himachal Pradesh, Northwestern Himalaya. Asian J Adv Basic Sci 2(1):77–98
Sharma S, Popli D, Sharma N, Chaurasia OP, Sood H (2016) Effect of temperature on in vitro organogenesis of Rhodiola imbricataEDGEW. – a Medicinal Herb. World Jounal of Pharmacy and Pharmaceutical Sciences 5(12):1228–1243. https://doi.org/10.20959/wjpps201612-8266
Shen MM, Wang Q, Yu XN, Teixeira da Silva JA (2012) Micropropagation of herbaceous peony (Paeonia lactiflora Pall). Sci Hort 148:30–38
Singh PP, Suresh PS, Bora PS, Bhatt V, Sharma U (2022) Govanoside B, a new steroidal saponin from rhizomes of Trillium govanianum. Nat Prod Res 36(1):37–45. https://doi.org/10.1080/14786419.2020.1761360
Sul IW, Korban SS (1994) Effect of different cytokinins on axillary shoot proliferation and elongation of several genotypes of Sequoia sempervirens. In vitro Cell Dev Biol 30:131–135
Suzuki K, Kawano S (2010) Seed germination and dispersal strategy of Trillium apetalon (Trilliaceae): a typical temperate woodland perennial in Japan. Plant Species Biol 25:231–239. https://doi.org/10.1111/j.1442-1984.2010.00295.x
Tomsone S, Gertnere D, Novikova D (2004) The influence of thidiazuron on shoot regeneration and proliferation of Rhododendrons in vitro. Acta Universitatis Latviensis Biology 676:239–242
Author Information
High Altitude Plant Physiology Research Center, H.N.B. Garhwal University, Srinagar Garhwal, India