Phytochemical profiling and bioactive assessment of Crassocephalum crepidiodes using GC-MS and LC-MS analysis
*Article not assigned to an issue yet
Research Articles | Published: 22 April, 2026
First Page: 0
Last Page: 0
Views: 422
Keywords: n Crassocephalum crepidioidesn , Antibiotic resistance, Antimicrobial, GC-MS, LC-MS
Abstract
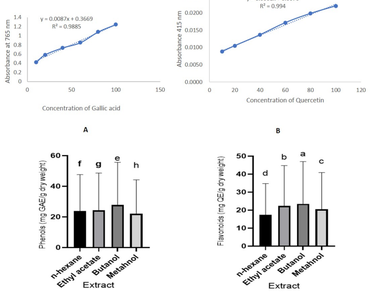
Crassocephalum crepidioides is an ethnomedicinal plant traditionally used for the treatment of various ailments and is widely consumed as a vegetable in certain parts of Northeast India and globally. The present study aims to determine the antioxidant and antimicrobial potential of extracts obtained from n-hexane, butanol, ethyl acetate, and methanol, and to identify the bioactive substances present in C. crepidioides. Preliminary phytochemical screening showed the presence of alkaloids, flavonoids, tannins, phytosterols, saponins, carbohydrates, and reducing sugars. The butanol extract exhibited the highest TPC (55.65 ± 0.008 mg/g) and TFC (46.98 ± 0.003 mg/g), as well as the highest antioxidant potential, with values of 27.04 ± 0.993 mg/mL and 24.9 ± 2.379 mg/mL against DPPH and ABTS free radicals, respectively. Furthermore, the butanol extract also showed the best antimicrobial activity. Since butanol extract showed high bioactive potential, it was chosen for further phytochemical analysis. Approximately six different classes of compounds were detected by GC-MS analysis, and ten different classes of compounds by LC-MS analysis. Our findings suggest that C. crepidioides is a promising therapeutic agent for a range of biomedical applications due to its strong antibacterial and antioxidant properties, as well as its numerous bioactive components. Thus, the present study provides a useful framework for further research into the functional roles and bioactivities of these compound classes.
References
Abdon JEO, Bautista JMDM, Omana ANA, Sajonia AV, Saldo IJP (2024) Phytochemical, antibacterial, and cytotoxicity analyses of redflower ragleaf (Crassocephalum crepidioides) leaves liquid extract. Sci Int (Lahore) 36(2):77–85
Adjatin A, Dansi A, Badoussi E et al. (2013) Phytochemical screening and toxicity studies of Crassocephalum rubens and Crassocephalum crepidioides consumed as vegetable in Benin. J Chem Pharm Res 5(6):160–167
Ahmad B, Khan MR, Jamil A, Ahmad MZ, Pervez S, Claridge TDW, Odell B, McCullagh JSO (2024) Antioxidant and antimicrobial potential of compounds isolated from Carissa opaca. Food Bioscien 57:103556. https://doi.org/10.1080/14786419.2010.529077
Ajuru MG, Williams LF, Ajuru G (2017) Qualitative and quantitative phytochemical screening of some plants used in ethnomedicine in the Niger Delta region of Nigeria. J Food Nutr Sci 5(5):198–205 https://doi.org/10.11648/j.jfns.20170505.16
Andrews JM (2001) Determination of minimum inhibitory concentrations. J Antimicrob Chemother 48(1):5–16. https://doi.org/10.1093/jac/48.suppl_1.5
Arawande JO, Komolafe EA, Imokhuede B (2013) Nutritional and phytochemical compositions of fireweed (Crassocephalum crepidioides). J Agric Technol 9(2):439–444. https://www.cabidigitallibrary.org/doi/pdf/https://doi.org/10.5555/20133105769
Badmus ANA, Schramm S, Gigl M, Iwebema W, Albertos P, Dawid C, Sieberer T, Poppenberger B (2022) Species-specific variation in abscisic acid homeostasis and responses impacts important traits in Crassocephalum orphan crops. Front Plant Sci 13:1–12. https://doi.org/10.3389/fpls.2022.923421
Bae JY, Seo YH, Oh SW (2022) Antibacterial activities of polyphenols against foodborne pathogens and their application as antibacterial agents. Food Sci Biotechnol 31(8):985–997. https://doi.org/10.1007/s10068-022-01058-3
Bahar E, Siddika MS, Nath B, Yoon H (2016) Evaluation of in vitro antioxidant and in vivo antihyperlipidemic activities of methanol extract of aerial part of Crassocephalum crepidioides (Asteraceae) Benth S Moore. Trop J Pharm Res 15(3):481–488. https://doi.org/10.4314/tjpr.v15i3.8
Baliyan S, Mukherjee R, Priyadarshini A, Vibhuti A, Gupta A, Pandey RP, Chang CM (2022) Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 27(4):1–15. https://doi.org/10.3390/molecules27041326
Bauer AW, Kirby WM, Sherris JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol 45(4):493–496. https://doi.org/10.1084/jem.20031325
Bello OM, Abiodun BO, Uduma AU (2019) Antioxidant and lipoxygenase inhibitory activity of Crassocephalum crepidioides (Benth.) S. Moore; an underutilized vegetable from Nigeria. Front Res Sci Crop Sci 1(2):28–31
Bello OM, Nok EA, Musa B, Musharaf A, Abiodun OB (2023) LC-MS analysis, total polyphenol content and antioxidant activity of ethanolic fractions of skin (epicarp) and seed pulp (mesocarp) of green and red wild Africa berries. https://doi.org/10.21203/rs.3.rs-3226316/v1
Bhandary SK, Bhat VS, Sharmila KP, Bekal MP (2012) Preliminary phytochemical screening of various extracts of Punica granatum peel, whole fruit and seeds. J Health Allied Sci NU 2(4):34–38. https://doi.org/10.1055/s-0040-1703609
Braca A, De Tommasi N, Di Bari L, Pizza C, Politi M, Morelli I (2001) Antioxidant principles from Bauhinia tarapotensis. J Nat Prod 64(7):892–895. https://doi.org/10.1021/np0100845
Can NM, Thao DTP (2020) Wound healing activity of Crassocephalum crepidioides (Benth.) S. Moore leaf hydroethanolic extract. Oxid Med Cell Longev 2020(1):1–10. https://doi.org/10.1155/2020/2483187
Chaachouay N, Zidane L (2024) Plant-derived natural products: a source for drug discovery and development. Drugs Drug Candidates 3(1):184–207. https://doi.org/10.3390/ddc3010011
Chang CC, Yang MH, Wen HM, Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal 10(3):102–106
Devi YA, Gnanasekaran P, Devi HJ (2024) Antibacterial, antioxidant and cytotoxicity assessment of Crassocephalum crepidioides leaf extract. J Pure Appl Microbiol 18(4):2528–2538. https://doi.org/10.22207/JPAM.18.4.24
Devi YA, Gnanasekaran P, Devi HJ, Siva D, Seshadri S, Ravindrran MB, Partheeban S, Bhuvaneswari S (2025) Antibiogram and antibacterial activity of Crassocephalum crepidioides (Thickhead) leaf extract against the wound isolates. J Appl Pharm Sci 15(09):041–051
Gokhale MS, Kokate CK, Purohit AP (2017) Pharmacog; Chap. 4:4.9–4.16
Gulluce M, Aslan A, Sokmen MU, Sahin FI, Adiguzel A, Agar G, Sokmen AT (2006) Screening the antioxidant and antimicrobial properties of the lichens Parmelia saxatilis, Platismatia glauca, Ramalina pollinaria, Ramalina polymorpha and Umbilicaria nylanderiana. Phytomed 13(7):515–521. https://doi.org/10.1016/j.phymed.2005.09.008
Ismaeel RO, Saliu BK, Usman LA, Hassan F, Adedeji OM, Akpan ED, Olanrewaju FSA (2025) Antibaterial activity of the GC-MS characterized essential oil from the leaves of Crassocephalum crepidiodes from North Central Nigeria. JEOPC 3(1):19–25
Jean BG, Irène FA, Donatien AA, Hervé-Hervé NA, Bertrand BM, Gael ADN, Merline NY, Franklik ZG, Bertrand DA, Théophile D (2022) Neuroprotective effects of the ethanolic leaf extract of Crassocephalum crepidioides (Asteraceae) on diazepam-induced amnesia in mice. Adv Pharmacol Pharm Sci 2022:1919469. https://doi.org/10.1155/2022/1919469
Joshi RK (2011) Terpene composition of Crassocephalum crepidioides from Western Ghats region of India. Int J Nat Prod Res 1(2):19–22. https://www.cabidigitallibrary.org/doi/full/https://doi.org/10.5555/20143410943
Kabir T, Samiha M, Yasmin H, Rahman S, Rahman S, Ashraf G, Akter R (2021) Free radical scavenging, thrombolytic and cytotoxic effects of the medicinal herb, Crassocephalum crepidioides. Res J Pharm Technol 14(4):2205–2210. https://doi.org/10.52711/0974-360X.2021.00391
Krishnamoorthy K, Subramaniam P (2014) Phytochemical profiling of leaf, stem and tuber parts of Solena amplexicaulis (Lam.) Gandhi using GC-MS. Int Sch Res Notices 2014:567409. https://doi.org/10.1155/2014/567409
Li X, Yuan W, Wu J, Zhen J, Sun Q, Yu M (2022) Andrographolide, a natural anti-inflammatory agent: an update. Front Pharmacol 13:920435. https://doi.org/10.3389/fphar.2022.920435
Murray BE (2000) Vancomycin-resistant enterococcal infections. N Engl J Med 342(10):710–721. https://doi.org/10.1056/NEJM200003093421007
Nwobodo CD, Ugwu MC, Anie OC, Al-Ouqaili MT, Ikem JC, Chigozie UV, Saki M (2022) Antibiotic resistance: the challenges and some emerging strategies for tackling a global menace. J Clin Lab Anal 36(9):e24655. https://doi.org/10.1002/jcla.24655
Omotehinwa FH, Ilesanmi OS, Oyegoke DA, Olagunju VA, Lajide L (2023) Comparative GC-MS profile for bioactive compounds of foliar surface extracts of Senecio biafrae and Crassocephalum crepidioides. Int j agric biol 30(3):215–220. https://doi.org/10.17957/IJAB/15.2078
Pastorino G, Cornara L, Soares S, Rodrigues F, Oliveira MBPP (2018) Liquorice (Glycyrrhiza glabra): a phytochemical and pharmacological review. Phytother Res 32(12):2323–2339. https://doi.org/10.1002/ptr.6178
Persson C, Robert E, Carlsson E et al. (2015) The effect of unsaturated fatty acid and triglyceride oil addition on the mechanical and antibacterial properties of acrylic bone cements. J Biomater Appl 30(3):279–289. https://doi.org/10.1177/0885328215581316
Riaz M, Khalid R, Afzal M, Anjum F, Fatima H, Zia S, Rasool G, Egbuna C, Mtewa AG, Uche CZ, Aslam MA (2023) Phytobioactive compounds as therapeutic agents for human diseases: a review. Food Sci Nutr 11(6):2500–2529. https://doi.org/10.1002/fsn3.3308
Sankara Rao K, Deepak K (2024) India Flora Online
Santhi K, Sengottuvel R (2016) Qualitative and quantitative phytochemical analysis of Moringa concanensis Nimmo. Int J Curr Microbiol Appl Sci 5(1):633–640. https://doi.org/10.20546/ijcmas.2016.501.064
Schramm S, Rozhon W, Adedeji-Badmus AN, Liang Y, Nayem S, Winkelmann T, Poppenberger B (2021) The orphan crop Crassocephalum crepidioides accumulates the pyrrolizidine alkaloid jacobine in response to nitrogen starvation. Front Plant Sci 12:1–13. https://doi.org/10.3389/fpls.2021.702985
Seal T, Chaudhuri K (2014) Ethnobotanical importance and nutritional potential of wild edible fruits of Meghalaya state in India. J Chem Pharm Res 6(10):680–684. https://www.cabidigitallibrary.org/doi/full/https://doi.org/10.5555/20143410943
Shaaban MT, Ghaly MF, Fahmi SM (2021) Antibacterial activities of hexadecenoic acid methyl ester and green-synthesized silver nanoparticles against multidrug-resistant bacteria. J Basic Microbiol 61(6):557–568. https://doi.org/10.1002/jobm.202100061
Singh G, Passari AK, Leo VV, Mishra VK, Subbarayan S, Singh BP, Kumar B, Kumar S, Gupta VK, Lalhlenmawia H, Nachimuthu SK (2016) Evaluation of phenolic content variability along with antioxidant, antimicrobial, and cytotoxic potential of selected traditional medicinal plants from India. Front Plant Sci 7:1–17. https://doi.org/10.3389/fpls.2016.00407
Sintupachee S, Sangphech N, Promden W (2025) Assessing the antioxidant activity and anti-inflammatory potency on lipopolysaccharide-induced RAW 264.7 macrophages of eleven methanolic extracts indigenous vegetables. ASEAN J Sci Technol Rep 28(2):e255909. https://doi.org/10.55164/ajstr.v28i2.255909
Sultana B, Anwar F, Ashraf M (2009) Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules 14(6):2167–2180. https://doi.org/10.3390/molecules14062167
Thoudam S, Sharma D (2020) A review study on nutraceutical and nutritional efficacy of Crassocephalum crepidioides. In: Hussain M (ed) Research Trends in Medicinal Plant Sciences. AkiNik Publications, New Delhi, India, pp 53–63
Threlfall EJ (2002) Antimicrobial drug resistance in Salmonella: problems and perspectives in food- and water-borne infections. FEMS Microbiol Rev 26(2):141–148. https://doi.org/10.1111/j.1574-6976.2002.tb00606.x
Tohma H, Gülçin İ, Bursal E, Gören AC, Alwasel SH, Köksal E (2017) Antioxidant activity and phenolic compounds of ginger (Zingiber officinale Rosc.) determined by HPLC–MS/MS. J Food Meas Charact 11(2):556–566. https://doi.org/10.1007/s11694-016-9423-z
Wijaya S, Nee TK, Jin KT, Din WM, Wiart C (2011) Antioxidant, anti- inflammatory, cytotoxicity and cytoprotection activities of Crassocephalum crepidioides (Benth.) S. Moore. Extracts and its phytochemical composition. Eur J Sci Res 67(1):157–165
Author Information
Department of Life Sciences, Pachhunga University College, Mizoram University, Aizawl, India