Microbial diversity associated with the natural spring water of Western Himalayas
Oza Yukti, Karande Mrunal, Ghosh Rochita, Rawat Janhvi Mishra, Purohit Sumit, Rawat Balwant, Zumla Alimuddin, Sharma Avinash, Karande Mrunal, Ghosh Rochita
Research Articles | Published: 10 June, 2024
First Page: 1795
Last Page: 1803
Views: 3160
Keywords: Western himalayas, Water springs, Targeted metagenomics, 16S rRNA gene, Fungal communities
Abstract
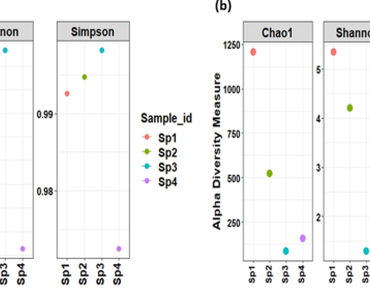
Springs in the Himalayan landscape are vital sources of potable water for millions of inhabitants who use it for domestic and farming purposes. The rise in urbanization, pilgrimage and various other anthropogenic activities invariably result in microbial contamination. In this study, we conducted a microbiological examination of water samples collected from four distinct Himalayan springs, located in the Rudraprayag district of Uttarakhand. We employed targeted metagenomics, using 16S rRNA and Internal Transcribed Spacer (ITS) gene analysis, to elucidate the bacterial and fungal composition. Cumulatively, the high-throughput sequencing data from all four springs revealed the abundance of Proteobacteria (35.38%), followed by Fermicutes (22.47%), Acidobacteriota (14.73%), and Actinobacteriota (5.35%). Genus level analysis of bacterial communities revealed the prevalence of Tumebacillus (13.85%) and Massilia (12.97%). Mycobiome analysis demonstrated the dominance of the fungal phyla Ascomycota (72.11%), followed by Basidiomycota (16.04%), Mortierellomycota (9.21%), and Chytridiomycota (1.2%). We further analysed the microbial communities using marker gene (16S rRNA) data through PICRUSt and the KEGG database to identify the gene abundance associated with carbon metabolism.
References
Kessy A, Allan Z, Timo P, Raivo P, Filipp I, Henrik RN, Urmas K (2023): Full UNITE + INSD dataset for eukaryotes. UNITE Community. (Accessed 2 December 2023) https://doi.org/10.15156/BIO/2938066
Babič MN, Gunde-Cimerman N, Vargha M, Tischner Z, Magyar D, Veríssimo C, Sabino R, Viegas C, Meyer W, Brandão J (2017) Fungal contaminants in drinking water regulation? A tale of ecology, exposure, purification and clinical relevance. Int J Environ Res Public Health 14(6). https://doi.org/10.3390/ijerph14060636
Begmatov S, Beletsky AV, Dedysh SN, Mardanov AV, Ravin NV (2022) Genome analysis of the candidate phylum MBNT15 bacterium from a boreal peatland predicted its respiratory versatility and dissimilatory iron metabolism. Frontiers in Microbiology, 13. https://doi.org/10.3389/fmicb.2022.951761
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA, Holmes SP (2016) May 23). DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13(7):581–583. https://doi.org/10.1038/nmeth.3869
Chaudhari D, Dhotre D, Jani K, Sharma A, Singh Y, Shouche YS, Rahi P (2020) Bacterial communities Associated with the Biofilms formed in high-Altitude Brackish Water Pangong Tso located in the Himalayan Plateau. Curr Microbiol 77(12):4072–4084. https://doi.org/10.1007/s00284-020-02244-4
Chauhan JS, Badwal T, Badola N (2020) Assessment of potability of spring water and its health implication in a hilly village of Uttarakhand, India. Appl Water Sci 10:73. https://doi.org/10.1007/s13201-020-1159-6
Chee-Sanford JC, Tian DH, Sanford RA (2019) Consumption of N2O and other N-cycle intermediates by Gemmatimonas aurantiaca strain T-27. Microbiology 165(12):1345–1354. https://doi.org/10.1099/mic.0.000847
Chhimwal M, Kaur S, Srivastava R, Hagare D, Prasad HJS (2022), April 1 Water quality of springs and lakes in the Kumaon Lesser Himalayan Region of Uttarakhand, India. Journal of Water and Health. https://doi.org/10.2166/wh.2022.028
Das S, Najar IN, Sherpa MT, Kumar S, Sharma P, Mondal K, Tamang S, Thakur N (2023) Baseline metagenome-assembled genome (MAG) data of Sikkim Hot Springs from Indian himalayan geothermal belt (IHGB) showcasing its potential CAZymes, and sulphur-nitrogen metabolic activity. World J Microbiol Biotechnol Incorporating MIRCEN J Appl Microbiol Biotechnology/World J Microbiol Biotechnol 39(7). https://doi.org/10.1007/s11274-023-03631-2
Devi SP, Jani K, Sharma A, Jha DK (2021) Bacterial communities and their bioremediation capabilities in oil-contaminated agricultural soils. Environ Monit Assess 194(1). https://doi.org/10.1007/s10661-021-09669-9
Douglas GM, Maffei VJ, Zaneveld J, Yurgel SN, Brown JR, Taylor CM, Huttenhower C, Langille MGI (2020) PICRUSt2 for prediction of metagenome functions. Nat Biotechnol 38(6):685–688. https://doi.org/10.1038/s41587-020-0548-6. (Accessed 10 December 2023)
Duan JL, Sun JW, Ji MM, Ma Y, Cui ZT, Tian RK, Xu PC, Sun WL, Yuan XZ (2020) Indicatory bacteria and chemical composition related to sulphur distribution in the river-lake systems. Microbiol Res 236(December 2019):126453. https://doi.org/10.1016/j.micres.2020.126453
Egidi E, Delgado-Baquerizo M, Plett JM, Wang J, Eldridge DJ, Bardgett RD, Maestre FT, Singh BK (2019) May 30). A few Ascomycota taxa dominate soil fungal communities worldwide. Nat Commun 10(1). https://doi.org/10.1038/s41467-019-10373-z
Figueroa-González I, Quijano G, Laguna IG, Muñoz R, García‐Encina PA (2016) A fundamental study on biological removal of N2O in the presence of oxygen. Chemosphere 158:9–16. https://doi.org/10.1016/j.chemosphere.2016.05.046
Green SJ, Prakash O, Jasrotia P, Overholt WA, Cardenas E, Hubbard D, Tiedje JM, Watson DB, Schadt CW, Brooks SC, Kostka JE (2012) Denitrifying Bacteria from the Genus Rhodanobacter dominate bacterial communities in the highly contaminated subsurface of a Nuclear Legacy Waste Site. Appl Environ Microbiol 78(4):1039–1047. https://doi.org/10.1128/aem.06435-11
Gupta A, Dutt V, Sharma N, Kajale S, Bhatt A, Shafi S, Azhar EI, Zumla A, Sharma A (2023) Examining the microbial composition of natural springs in Bhaderwah, Jammu and Kashmir, India. Environ Monit Assess 195(8). https://doi.org/10.1007/s10661-023-11507-z
Gupta S, Pandotra P, Gupta AP, Dhar JK, Sharma G, Ram G, Husain MK, Bedi YS (2010) Volatile (as and hg) and non-volatile (Pb and cd) toxic heavy metals analysis in rhizome of Zingiber officinale collected from different locations of North Western Himalayas by Atomic absorption spectroscopy. Food Chem Toxicol 48(10):2966–2971. https://doi.org/10.1016/j.fct.2010.07.034
Halbwachs H, Karasch P, Griffith G (2013) The diverse habitats of hygrocybe– peeking into an enigmatic lifestyle. Mycosphere 4(4):773–792. https://doi.org/10.5943/mycosphere/4/4/14
Jani K, Bandal J, Shouche YS, Shafi S, Azhar EI, Zumla A, Sharma A (2021) Extended ecological restoration of bacterial communities in the Godavari River during the COVID-19 Lockdown Period: a spatiotemporal Meta-analysis. Microb Ecol 82(2):365–376. https://doi.org/10.1007/s00248-021-01781-0
Jani K, Mahajan AS, Kajale S, Ashar A, Sharma A (2022) Soil bacterial community structure, metabolic adaptations and their functional interactions to abiotic factors in Antarctica. Pol Polar Res. https://doi.org/10.24425/ppr.2021.138588
Jiang C, Liu Y, Liu Y, Guo X, Liu SJ (2009) Isolation and characterization of ferrous- and sulphur-oxidizing bacteria from Tengchong solfataric region, China. J Environ Sci 21(9):1247–1252. https://doi.org/10.1016/S1001-0742(08)62411-0
Jiya N, Shede P, Sharma A (2023) Diversity and composition of fungal communities across diverse environmental niches in Antarctica. Polar Sci 38:100973. https://doi.org/10.1016/j.polar.2023.100973
Kumar M, Kalra N, Singh H, Sharma S, Singh Rawat P, Kumar Singh R, Kumar Gupta A, Kumar P, Ravindranath NH (2021) Indicator-based vulnerability assessment of forest ecosystem in the Indian Western Himalayas: an analytical hierarchy process integrated approach. Ecol Ind 125(March):107568. https://doi.org/10.1016/j.ecolind.2021.107568
Li M, Zhang Q, Sun X, Ka., Zeng C, Pandey A, Rawat B, Zhang F (2020) Heavy metals in surface sediments in the trans-himalayan Koshi River catchment: distribution, source identification and pollution assessment. Chemosphere 244. https://doi.org/10.1016/j.chemosphere.2019.125410
Mahato NK, Sharma A, Singh Y, Lal R (2019) Comparative metagenomic analyses of a high-altitude himalayan geothermal spring revealed temperature-constrained habitat-specific microbial community and metabolic dynamics. Arch Microbiol 201(3):377–388. https://doi.org/10.1007/s00203-018-01616-6
Martin (2023), October 19 Water and Sanitation - United Nations Sustainable Development. United Nations Sustainable Development. https://www.un.org/sustainabledevelopment/water-and-sanitation/
Mehrani MJ, Sobotka D, Kowal P, Ciesielski S, Makinia J (2020) The occurrence and role of Nitrospira in nitrogen removal systems. Bioresour Technol 303(November 2019). https://doi.org/10.1016/j.biortech.2020.122936
McMurdie PJ, Holmes S (2013), April 22 phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE, 8(4), e61217. https://doi.org/10.1371/journal.pone.0061217 (Accessed 2 December 2023)
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’hara RB, Oksanen MJ (2013) Package ‘vegan’. Community Ecol Package Version 2(9):1–295 (Accessed 2 December 2023)
Oliveros JC (2007–2015) Venny. An Interactive Tool for Comparing Lists with Venn’s Diagrams. https://bioinfogp.cnb.csic.es/tools/venny/index.html. (Accessed 14 December 2023)
Panwar S (2020) Vulnerability of himalayan springs to climate change and anthropogenic impact: a review. J Mt Sci 17:117–132. https://doi.org/10.1007/s11629-018-5308-4
Park D, Kim H, Yoon S (2017) Nitrous Oxide reduction by an Obligate Aerobic Bacterium, Gemmatimonas aurantiaca strain T-27. Appl Environ Microbiol 83(12). https://doi.org/10.1128/aem.00502-17
Pang Y, Wang J, Li S, Ji G (2021) Long-term sulfide input enhances chemoautotrophic denitrification rather than DNRA in freshwater lake sediments. Environ Pollut 270:116201. https://doi.org/10.1016/j.envpol.2020.116201
Perincherry L, Lalak-Kańczugowska J, Stępień Ł (2019) Fusarium-produced mycotoxins in Plant-Pathogen interactions. Toxins 11(11):664. https://doi.org/10.3390/toxins11110664
Qayoom U, Islam ST, Sabha I, Bhat SU, Dar SA (2022) Coliform pollution mapping in major watersheds along Jhelum River Basin of Kashmir Himalaya. Environ Sci Pollut Res 30(3):7930–7941. https://doi.org/10.1007/s11356-022-22727-0
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, Peplies J, Glöckner FO (2012), November 27 The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Research, 41(D1), D590–D596. https://doi.org/10.1093/nar/gks1219 (Accessed 2 December 2023)
Rambia A, Veluchamy C, Rawat J, Jamdhade MD, Purohit S, Pawar KD, Chandrasekaran R, Rawat B, Sharma A (2023) Revealing bacterial and fungal communities of the untapped forest and alpine grassland zones of the Western-Himalayan region. https://doi.org/10.1007/s10123-023-00430-5. International Microbiology
Rashid I, Romshoo SA (2013) Impact of anthropogenic activities on water quality of Lidder River in Kashmir Himalayas. Environ Monit Assess 185:4705–4719. https://doi.org/10.1007/s10661-012-2898-0
Rincón-Tomás B, Lanzén A, Sánchez P, Estupiñán M, Sanz-Sáez I, Bilbao ME, Rojo D, Mendibil I, Pérez-Cruz C, Ferri MG, Capo E, Abad-Recio IL, Amouroux D, Bertilsson S, Sánchez O, Acinas SG, Alonso-Sáez L (2023) Revisiting the mercury cycle in marine sediments: a potential multifaceted role for Desulfobacterota. J Hazard Mater 133120. https://doi.org/10.1016/j.jhazmat.2023.133120
Sati SC, Paliwal PC (2008) Physico-chemical and bacteriological analysis of Kosi River water in central Himalaya. Pollution Res 27(1):179–183
Sharma A, Jani K, Shouche YS, Pandey A (2014) Microbial diversity of the Soldhar hot spring, India, assessed by analyzing 16S rRNA and protein-coding genes. Ann Microbiol 65(3):1323–1332. https://doi.org/10.1007/s13213-014-0970-4
Sharma A, Karande M, Zumla A (2023a) Advancing sewage surveillance at mass gathering events for reducing transmission of antimicrobial resistant bacterial pathogens. Travel Med Infect Dis 54:102619. https://doi.org/10.1016/j.tmaid.2023.102619
Sharma A, Rodriguez-Morales AJ, Traore T, Shafi S, El-Kafrawi SA, Azhar EI, Zumla A (2023b) Globalisation of antibiotic-resistant bacteria at recurring mass gathering events. Lancet (London England) 402(10398):e5–e7. https://doi.org/10.1016/S0140-6736(22)01995-X
Sharma P, Kumar P, Sharma M, Butail N, Kumar P (2022) Vulnerability of Water resources to Climate Change in Indian Himalayan Region. Indian J Ecol 49. https://doi.org/10.55362/ije/2022/3816
Springer AE, Stevens LE (2009) Spheres of discharge of springs. Hydrogeol J 17:83–93. https://doi.org/10.1007/s10040-008-0341-y
Verma R, Jamwal P (2022) Sustenance of himalayan springs in an emerging water crisis. Environ Monit Assess 194(2). https://doi.org/10.1007/s10661-021-09731-6
Volk TJ (2013) Fungi. Encyclopedia of Biodiversity, (Second Edition), Academic Press, https://doi.org/10.1016/B978-0-12-384719-5.00062-9
Wang M, Tian J, Xiang M, Liu X (2017), July 3 Living strategy of cold-adapted fungi with the reference to several representative species. Mycology, 8(3), 178–188. https://doi.org/10.1080/21501203.2017.1370429
Wang X, Zhou Q, Zijing L, Xia L, Song S, Meza JVG, Montes ML, Li J (2024), January 1 Surge of native rare taxa in tailings soil induced by peat bacterial invasion. Science of the Total Environment.https://doi.org/10.1016/j.scitotenv.2023.168596
Wani PA, Rafi N, Wani U, A, H. B., Khan MSA (2021) Simultaneous bioremediation of heavy metals and biodegradation of hydrocarbons by metal resistant Brevibacillus parabrevis OZF5 improves plant growth promotion. Bioremediat J 27(1):20–31. https://doi.org/10.1080/10889868.2021.1989371
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York ([Internet], https://ggplot2.tidyverse.org/ (Accessed 2 December)
Yadav N, Rakholia S, Ali N, Yosef R (2023) Fungal diversity and distribution in the Biodiversity hotspots of the Western Himalayas. Diversity 15(11):1106. https://doi.org/10.3390/d15111106
Zhu B, Karwautz C, Andrei S, Klingl A, Pernthaler J, Lueders T (2022) A novel Methylomirabilota methanotroph potentially couples methane oxidation to iodate reduction. MLife 1(3):323–328. https://doi.org/10.1002/mlf2.12033
Author Information
National Centre for Microbial Resource, National Centre for Cell Science, Pune, India