Marine origin Bacillus subtilis 10MH17 as an effective bio-inoculant for improving growth of Vigna unguiculata plant
*Article not assigned to an issue yet
Research Articles | Published: 22 April, 2026
First Page: 0
Last Page: 0
Views: 247
Keywords: Marine microbe, Plant growth promoters, Cowpeas, Pot trials
Abstract
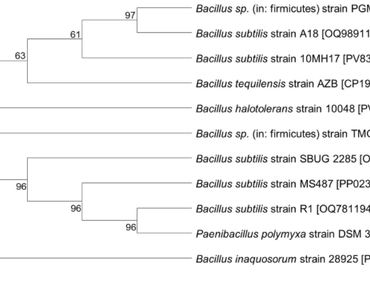
The rising global food demand has increased the need for sustainable strategies that enhance agricultural productivity. Among these, plant growth promoting bacteria (PGPB) are gaining importance for their ability to improve plant development, soil fertility, and crop resilience. While extensive research has focused on soil based PGPB, marine microorganisms remain comparatively underexplored. This study evaluates a marine derived Bacillus subtilis strain, 10MH17, for its plant growth promoting potential. The isolate demonstrated multiple beneficial traits, including ammonia production, phosphate and potassium solubilization, and effective root colonization in cowpea. A talc-based bioformulation of B. subtilis 10MH17 was prepared and tested through pot experiments under two conditions: control (T1) and treatment (T2). Plants treated with B. subtilis 10MH17 showed a 14.17% increase in biomass and a 16.2% increase in height over the control. Additionally, the treatment improved chlorophyll content, enhanced stress related biochemical responses, and elevated soil nitrogen, potassium, and phosphorus levels. Overall, the findings highlight marine B. subtilis 10MH17 as a promising PGPB for improving cowpea growth and soil health, supporting its potential role in sustainable agriculture.
References
Aleksandrov VG, Blagodyr RN, Ilev IP (1967) Liberation of phosphoric acid from apatite by silicate bacteris. Mikrobiolohichnyi Zhurnal (Kiev) 29:111–114
Anonymous. Directorate of Pulse Development (Bhopal) Annual Progress Report, Integrated Scheme of Oilseed, Pulses, Oil palm and Maize (ISOPOM) –Pulses; 2007
Ayalew T, Yoseph T (2022) Cowpea (Vigna unguiculata L. Walp.): a choice crop for sustainability during the climate change periods. J Appl Biol Biotechnol 10(3):154–162. https://doi.org/10.7324/JABB.2022.100320
Chhonkar PK, Subba-Rao NS (1967) Fungi associated with legume root nodules. Can J Microbiol 13(7):749–753. https://doi.org/10.1139/m67-099
Demutskaya LN, Kalinichenko IE (2010) Photometric determination of ammonium nitrogen with the Nessler reagent in drinking water after its chlorination. J Water Chem Technol 32(2):90–94. https://doi.org/10.3103/S1063455X10020049
Goswami D, Vaghela H, Parmar S, Dhandhukia P, Thakker JN (2013) Plant growth promoting potentials of Pseudomonas spp. strain OG isolated from marine water. J Plant Interact 8(4):281–290. https://doi.org/10.1080/17429145.2013.768360
Goswami D, Patel K, Parmar S, Vaghela H, Muley N, Dhandhukia P, Thakker JN (2015) Elucidating multifaceted urease producing marine Pseudomonas aeruginosa BG as a cogent PGPR and bio-control agent. Plant Growth Regul 75(1):253–263. https://doi.org/10.1007/s10725-014-9949-1
Hossain MT, Islam T (2022) Amelioration of salinity stress by bacillus species as promoters of plant growth in saline soil. In: Bacilli in climate resilient agriculture and bioprospecting, pp 199–208. https://doi.org/10.1007/978-3-030-85465-2_9
Kálmán CD, Nagy Z, Berényi A, Kiss E, Posta K (2023) Investigating PGPR bacteria for their competence to protect hybrid maize from the factor drought stress. Cereal Res Commun 52(1):129–150. https://doi.org/10.1007/s42976-023-00388-0
King JE (1932) The colorimetric determination of phosphorus. Biochem J 26:292–297. https://doi.org/10.1042/bj0260292
Martins SJ, Rocha GA, De Melo HC, De Castro Georg R, Ulhôa CJ, De Campos Dianese É, Oshiquiri LH, Da Cunha MG, Da Rocha MR, De Araújo LG, Vaz KS, Dunlap CA (2018) Plant-associated bacteria mitigate drought stress in soybean. Environ Sci Pollut Res 25(14):13676–13686. https://doi.org/10.1007/s11356-018-1610-5
Melo LHV, Rocha FYO, Vidal MS, Gitahy P, Arruda GM, Barreto CP, Alves PB, Ramos E, Rossi CN, Schwab S, Boa Sorte PMF, Avelar JP, Baldani JI (2021) Diversity and biotechnological potential of endophytic Bacillus species originating from the stem apoplast fuid of sugarcane plants. Appl Soil Ecol 166:103985. https://doi.org/10.1016/j.apsoil.2021.103985
Oren A (2008) Microbial life at high salt concentrations: phylogenetic and metabolic diversity. Saline Syst 4(1):2. https://doi.org/10.1186/1746-1448-4-2
Patel P, Patel K, Dhandhukia P, Thakker JN (2021) Plant growth promoting traits of marine Micrococcus sp. with bio-control ability against Fusarium in chickpea plant. Vegetos 34(1):94–101. https://doi.org/10.1007/s42535-021-00191-4
Rao S (1977) Soil microorganisms and plant growth
Rathod K, Rana S, Dhandukia P, Thakker JN (2023) Investigating marine Bacillus as an effective growth promoter for chickpea. J Genet Eng Biotechnol 21(1):137. https://doi.org/10.1186/s43141-023-00608-4
Rathod K, Rana S, Dhandhukia P, Thakker JN (2024) From sea to soil: marine Bacillus subtilis enhancing chickpea production through in vitro and in vivo plant growth promoting traits. Braz J Microbiol 55(1):823–836. https://doi.org/10.1007/s42770-023-01238-1
Riah W, Laval K, Laroche-Ajzenberg E, Mougin C, Latour X, Trinsoutrot-Gattin I (2014) Effects of pesticides on soil enzymes: a review. Environ Chem Lett 12(2):257–273. https://doi.org/10.1007/s10311-014-0458-2
Sharma S, Sayyed R, Trivedi M, Gobi T (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2(1):587. https://doi.org/10.1186/2193-1801-2-587
Soldan R, Mapelli F, Crotti E, Schnell S, Dafonchio D, Marasco R, Fusi M, Borin S, Cardinale M (2019) Bacterial endophytes of mangrove propagules elicit early establishment of the natural host and promote growth of cereal crops under salt stress. Microbiol Res 223–225:33–43. https://doi.org/10.1016/j.micres.2019.03.008
Thakker JN, Patel P, Dhandhukia PC (2011) Induction of defence-related enzymes in susceptible variety of banana: role of Fusarium derived elici - tors. Arch Phytopathol Plant Protect 44(20):1976–1984. https://doi.org/10.1080/03235408.2011.559032
Tsavkelova EA, Cherdyntseva TA, Klimova SY, Shestakov AI, Botina SG, Netrusov AI (2007) Orchid-associated bacteria produce indole-3-acetic acid, promote seed germination, and increase their microbial yield in response to exogenous auxin. Arch Microbiol 188(6):655–664. https://doi.org/10.1007/s00203-007-0286-x
Abhigna, M., Kulkarni, N. S., Channakeshava, R., & Sridhar, K. (2025). Population dynamics of cowpea aphid Aphis craccivora (Koch) in fodder cowpea Vigna unguiculata (L.) Walp. Journal of Farm Sciences, 38(1), 52–55
Saitou, N., & Nei, M. (1987). The neighbor-joining method: A new method for reconstructing phylogenetic trees. Molecular Biology and Evolution, 4(4), 406–425
Felsenstein, J. (1985). Confidence limits on phylogenies: An approach using the bootstrap. Evolution, 39(4), 783–791
Tamura, K., Nei, M., & Kumar, S. (2004). Prospects for inferring very large phylogenies by using the neighbor-joining method. Proceedings of the National Academy of Sciences, 101(30), 11030–11035
Author Information
Department of Biological Sciences, P.D. Patel Institute of Applied Sciences, CHARUSAT University, Changa, Anand, India