In vitro antioxidant and antimicrobial potentials of aqueous extract of Picralima nitida seeds
Adeola Oluwakemi E., Akpor Oluwaseyi A., Adamolekun Modupe M., Adewale Olusola B., Akpor Oghenerobor B.
Research Articles | Published: 11 December, 2023
First Page: 486
Last Page: 493
Views: 3472
Keywords: Antibacterial, Antioxidant, Bioactivity, Growth inhibition, P. nitida
Abstract
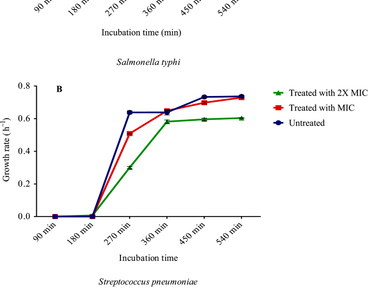
The seeds of Picralima nitida are a good source of vital minerals like zinc, iron, and manganese as well as amino acids, vitamins A and E. The objective of this study was to determine the phytochemical content, and assess the antioxidant and antimicrobial activities of aqueous extract of P. nitida seeds. Antibacterial assay of the extract was carried out against seven pathogens using the agar well diffusion procedure. The antioxidant capacity of the extract was evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) scavenging activity assay. Phytochemical composition of the extract was conducted utilizing quantitative as well as qualitative assay procedures while bioactive composition was done using gas chromatography-mass spectroscopy. From the results, the growth of Escherichia coli, Xanthomonas capestris, Pseudomonas aeruginosa, and Bacillus subtilis were not inhibited in presence of the extract. Staphylococcus aureus and Streptococcus pneumonia were both inhibited by a concentration of 30 mg/mL, and 10 mg/mL for Salmonella typhi. Generally, % inhibition of DPPH scavenging activity of the extract and control (quercetin) was observed to follow a concentration dependent manner. For almost all the concentrations investigated, % inhibition in the presence of the extract showed remarkable higher values than what was observed with the control. An examination of the extract’s phytochemical composition revealed the presence of alkaloids, saponins, flavonoids, phenols, and tannins in varying concentrations. With respect to active compounds present, the dominant compounds were Erucic acid, 9,12-Octadecadienoic acid (Z,Z), 9-Octadecenoic acid, methyl ester, n-Hexadecanoic acid, Linoleoyl chloride, Phenol and 3,5-bis (1,1-dimethyl ethyl). Based on the study findings, it could be inferred that the use of the plant part for possible development of pharmaceutical product is promising.
References
Aghedo ON, Owolabi JB, Ogbeide OK (2021) Chemical composition and antimicrobial activities of Picralima Nitida stem bark extracts. Chemsearch J 12(2):55–63
Akabassi GC, Padonou EA, Yao EJ, Nakpalo S, Palanga KK, Assogbadjo BE, Zandjanakou-Tachin M, Assogbadjo AE, Zirihi NG (2022) Extract yield, dilution methods and antifungal potential of fruits of Picralima nitida (Stapf.) TA Durand & H. Durand. J Saudi Soc Agric Sci 21(7):425 – 31. https://doi.org/10.1016/j.jssas.2021.11.006
Akpor OB, Ndakotsu J, Evbuomwan IO, Olaolu TD, Osemwegie OO (2021) Bacterial growth inhibition and antioxidant potentials of leaf infusions of (Moringa oleifera), Locust beans (Parkia biglobosa) and bitter leaf (Vernonia amygladina). Sci Afr 14:e01001. https://doi.org/10.1016/j.sciaf.2021.e01001
Aparna V, Dileep KV, Mandal PK, Karthe P, Sadasivan C, Haridas M (2012) Anti-inflammatory property of n‐hexadecanoic acid: structural evidence and kinetic assessment. Chem Biol Drug Des 80(3):434–439. https://doi.org/10.1111/j.1747-0285.2012.01418.x
Bruce SO, Onyegbule FA, Ihekwereme CP (2016) Evaluation of the hepato-protective and anti-bacterial activities of ethanol extract of Picralima Nitida seed and pod. J Phytomed Ther 15(2):1–22
Dapaah G (2016) Effect of the ethanol seed extract of Picralima nitida (stapf) TH. &H. Durand) on cough and its complications. Master of Sciences Thesis, Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
Dzotam JK, Kuete V (2023) Picralima Nitida as a potential source of antibacterial agents. Adv Bot Res 107:275–288
Egamberdieva D, Wirth SJ, Alqarawi AA, Abd_Allah EF, Hashem A (2017) Phytohormones and beneficial microbes: essential components for plants to balance stress and fitness. Front Microbiol 8:2104
Egbuna C, Ifemeje JC, Maduako MC, Tijjani H, Udedi SC, Nwaka AC, Ifemeje MO (2018) Phytochemical test methods: qualitative, quantitative and proximate analysis. Phytochemistry. Apple Academic Press, pp 381–426
Fakeye TO, Itiola OA, Odelola HA (2000) Evaluation of the antimicrobial property of the stem bark of Picralima Nitida (Apocynaceae). Phytother Res 14(5):368–370
Falodun A, Okunrobo LO, Uzoamaka N (2006) Phytochemical screening and anti-inflammatory evaluation of methanolic and aqueous extracts of Euphorbia heterophylla Linn (Euphorbiaceae). Afr J Biotechnol 5(6):529–531
Feyisayo AK, Victor AC (2019) Assessment of antioxidant and antidiabetic properties of Picralima Nitida seed extracts. J Med Plant Res 13(1):9–17. https://doi.org/10.5897/JMPR2018.6680
Harborne AJ (1998) Phytochemical methods a guide to modern techniques of plant analysis. Springer Science & Business Media
Ifemeje JC, Egbuna C, Udedi SC, Iheukwumere HI (2014) Phytochemical and in vitro antibacterial evaluation of the ethanolic extract of the stem bark of Entada Africana Guill. & Perr and Sarcocephalus Latifolus. Int J Biochem Res Rev 4(6):584
Inkoto CL, Kayembe JP, Mpiana PT (2020) A review on the phytochemistry and pharmacological properties of Picralima nitida Durand and H. (Apocynaceae family): A potential antiCovid-19 medicinal plant species. Emergent Life Sci Res 6:64–75. https://doi.org/10.31783/elsr.2020.616475
Kumar JS, Sharma B (2022) A review on neuropharmacological role of erucic acid: an omega-9 fatty acid from edible oils. Nutr Neurosci 25(5):1041–1055. https://doi.org/10.1080/1028415X.2020
Li S, Odedina S, Agwai I, Ojengbede O, Huo D, Olopade OI (2020) Traditional medicine usage among adult women in Ibadan, Nigeria: a cross-sectional study. BMC Complement Altern Med 20:1–7. https://doi.org/10.1186/s12906-020-02881-z
Merveille TO, Denis Z, Abdel NJ, Ursula AA, Adie AF, Smith BB, Clement AN (2017) Antimicrobial activities of some medicinal plants against multiresistants microorganisms causing urogenital tract Infections in Cameroon. J Dis Med Plants 3(2):33–41. https://doi.org/10.11648/j.jdmp.20170302.12
Nwankwo Nicodemus E, Nwodo Fred OC, Joshua Parker E (2017) Free radical scavenging property of Picralima Nitida seed extract on malaria-induced albino mice. Am J Life Sci 5(5):125–133. https://doi.org/10.11648/j.ajls.20170505.12
Nwaogu LA (2016) Chemical profile of Picralima Nitida seeds used in ethnomedicine in West Africa. Futo J Sci 2(2):110–122
Obasi NA, Ukadilonu J, Eze E, Akubugwo EI, Okorie UC (2012) Proximate composition, extraction, characterization and comparative assessment of coconut (Cocos nucifera) and melon (Colocynthis citrullus) seeds and seed oils. Pak J Biol Sci 15(1):1–9. https://doi.org/10.3923/pjbs.2012.1.9
Odoh AE, Yéhé DM, Kanga Y, Zirihi GN, Koné-Bamba D (2021) Comparative study of the antioxidant activity of Holarrhena floribunda and Picralima Nitida. Turk J Agric: Food Sci Technol 9(10):1925–1930. https://doi.org/10.24925/turjaf.v9i10.1925-1930.4514
Okonta JM, Aguwa CN (2007) Evaluation of hypoglycemic activity of glycosides and alkaloids extracts of Picralima Nitida Stapf (Apocynaceae) seed. Int J Pharmacol 3(6):505–509. https://doi.org/10.3923/ijp.2007.505.509
Okoronkwo I, Onyia-Pat JL, Okpala P, Agbo MA, Ndu A (2014) Patterns of complementary and alternative medicine use, perceived benefits, and adverse effects among adult users in Enugu Urban, Southeast Nigeria. J Evid Based Complementary Altern Med 2014: 239372 https://doi.org/10.1155/2014/239372
Poudyal H, Panchal SK, Diwan V, Brown L (2011) Omega-3 fatty acids and metabolic syndrome: effects and emerging mechanisms of action. Prog Lipid Res 50(4):372–387. https://doi.org/10.1016/j.plipres.2011.06.003
Shirwaikar A, Shirwaikar A, Rajendran K, Punitha IS (2006) In vitro antioxidant studies on the benzyl tetra isoquinoline alkaloid berberine. Biol Pharm Bull 29(9):1906–1910. https://doi.org/10.1248/bpb.29.1906
Sofowora A (1993) Medicinal Plant and Traditional Medicine in Africa. Chichester John
Author Information
Faculty of Nursing Sciences, Afe Babalola University, Ado-Ekiti, Nigeria