In silico investigation and MD simulations of phytochemicals of C. wightii against dengue targets NS5 and E protein
Research Articles | Published: 06 July, 2023
First Page: 1166
Last Page: 1184
Views: 3573
Keywords: Docking, n Commiphora wighttin , Toxicity, Drug likeness, MD simulation
Abstract
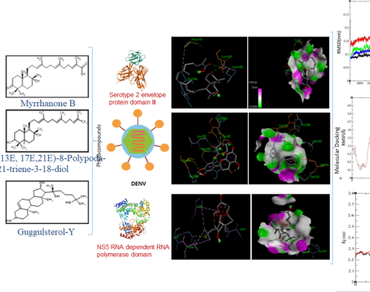
Dengue fever continues to be a global issue which does not have a specific remedy. Many different types of studies have recently been carried out in order to find a potential treatment for the dengue virus using natural resources. Commiphora wightti plant is also one such medicinal plants that has been reported to have potential antiviral activity in the treatment of many viral diseases. An in silico molecular binding study is conducted on a library of 52 bioactive compounds of Commiphora wightti against dengue virus protein targets, E protein (PDB ID: 3UZV) and NS5 methyl transferase (PDB ID: 2J7U). The molecular docking results showed that the Commipherin (− 8.2 kcal/mol) and Myrrhanone B (− 8.0 kcal/mol) have excellent binding affinity with NS5 methyl transferase while Myrrhanone A acetate (− 11.8 kcal/mol) and Myrrhanone B (− 11.1 kcal/mol) showed it for E-protein target. From the best ten selected phytoconstituents, three have followed the Lipinski’s rule and showed good drug likeness score and also satisfied all the ADME and toxicity analysis criteria, viz. Myrrhanone B (14), (13E, 17E,21E)-8-Polypoda-13,17,21-triene-3-18-diol (22) and Guggul sterol- Y (37) which were persuaded for Molecular dynamic simulations to find out new potential drug candidate against DENV. Stability of the Myrrhanone B (14)-NS5 complex was found maximum with RMSD value of 0.2nm. But on the basis of RMSF, Rg and no. of hydrogen bonds, molecule no. 37 (Guggulsterol-Y) and 22 ((13E, 17E,21E)-8-Polypoda-13,17,21-triene-3-18-diol) were found most suitable NS5 inhibitor with considerable RMSD. Rg values of NS5-22 and NS5-37 is scattered between 2.2 nm to 2.3 nm. These phytochemicals have shown significant potential of a therapeutic in In-silico studies and further in vitro and in vivo validation of the results is needed in view of finding potential therapeutic agent against Dengue Virus.
Graphical Abstract

References
Ahmad RS, Hussain MB, Sultan MT, Arshad MS, Waheed M, Shariati MA, Plygun S, Hashempur MH (2020) Biochemistry, safety, pharmacological activities, and clinical applications of turmeric: a mechanistic review. Evid-based Complement Altern Med. https://doi.org/10.1155/2020/7656919
Bhardwaj M, Alia A (2019) Commiphora wightii (Arn.) Bhandari. Review of its botany, medicinal uses, pharmacological activities and phytochemistry. J Drug Deliv Ther. https://doi.org/10.22270/jddt.v9i4-s.3256
Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF (2013) The global distribution and burden of dengue. Nature. https://doi.org/10.1038/nature12060
De A, Ray HP, Jain P, Kaur H, Singh N (2020) Synthesis, characterization, molecular docking and DNA cleavage study of transition metal complexes of o-vanillin and glycine derived Schiff base ligand. J Mol Struct 1199:126901. https://doi.org/10.1016/j.molstruc.2019.126901
De Paula SO, Fonseca BALD (2004) Dengue: a review of the laboratory tests a clinician must know to achieve a correct diagnosis. Brazilian J Infect Dis 8:390–398. https://doi.org/10.1590/S1413-86702004000600002
Dwivedi VD, Tripathi IP, Bharadwaj S, Kaushik AC, Mishra SK (2016) Identification of new potent inhibitors of dengue virus NS3 methyl transferase from traditional Chinese medicine database. Virusdisease 27(3):220–225. https://doi.org/10.1007/s13337-016-0328-6
Gkg MG, Kouri G (2002) Dengue: an update. Lancet Infect Dis. https://doi.org/10.1016/s1473-3099(01)00171-2
Guzman MG, Halstead SB, Artsob H, Buchy P, Farrar J, Gubler DJ, Hunsperger E, Kroeger A, Margolis HS, Martínez E, Nathan MB (2010) Dengue: a continuing global threat. Nat Rev Microbiol 8(12):S7–S16. https://doi.org/10.1038/nrmicro2460
Halstead SB (2007) Dengue. Lancet 370(9599):1644–1652. https://doi.org/10.1016/S0140-6736(07)61687-0
Jain P, Singh V, Ali S, Tripathi V, Saraswat U (2018) Synthesis, characterization, molecular docking and biological activity of 5, 6-bis-(4-fluoro-phenyl)-3, 4, 7, 8-tetraaza-bicyclo [8.3. 1] tetradeca-1 (13), 4, 6, 10 (14), 11-pentaene-2, 9-dione and its transition metal complexes. J Saudi Chem Soc 22(5):546–557. https://doi.org/10.1016/j.jscs.2017.09.005
Jain P, Guin M, De A, Singh M (2022) Molecular docking, synthesis, anticancer activity and computational investigations of thiazole based ligands and their Cu (II) complexes. J Phys Org Chem. https://doi.org/10.1002/poc.4384
Jarerattanachat V, Boonarkart C, Hannongbua S, Auewarakul P, Ardkhean R (2023) In silico and in vitro studies of potential inhibitors against Dengue viral protein NS5 Methyl Transferase from Ginseng and Notoginseng. J Tradit Complement Med. https://doi.org/10.1016/j.jtcme.2022.12.002
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand–protein interaction diagrams for drug discovery. J Chem Inf Model. https://doi.org/10.1021/ci200227u
Lim SP, Noble CG, Shi PY (2015) The dengue virus NS5 protein as a target for drug discovery. Antiviral Res 119:57–67. https://doi.org/10.1016/j.antiviral.2015.04.010
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J Comput Chem 30(16):2785–2791. https://doi.org/10.1002/jcc.21256
Paul A, Raj AVVS (2021) Evaluation of antiviral activity of Andrographis paniculata and Tinospora cordifolia using in silico and in vitro assay against DENV-2. J Pharmacogn Phytochem 10(2):486–496. https://doi.org/10.22271/phyto.2021.v10.i2f.13847
Powers N, C. and, Setzer N, W (2016) An in-silico investigation of phytochemicals as antiviral agents against dengue fever. Comb Chem High Throughput Screen 19(7):516–536. https://doi.org/10.2174/1386207319666160506123715
Rahman MM, Biswas S, Islam KJ, Paul AS, Mahato SK, Ali MA, Halim MA (2021) Antiviral phytochemicals as potent inhibitors against NS3 methyl transferase of dengue virus. Comput Biol Med 134:104492. https://doi.org/10.1016/j.compbiomed.2021.104492
Rani R, Mishra S (2013) Phytochemistry of guggul (Commiphora wightii): A review. Asian J Res Chem 6(4):415. https://doi.org/10.5958/0974-4150
Reza MN, Ferdous N, Emon M, Hossain T, Islam M, Mohiuddin AKM, Hossain MU (2021) Pathogenic genetic variants from highly connected cancer susceptibility genes confer the loss of structural stability. Sci Rep 11(1):1–19
Rolta R, Salaria D, Sharma P, Sharma B, Kumar V, Rathi B, Verma M, Sourirajan A, Baumler DJ, Dev K (2021) Phytocompounds of Rheum emodi, Thymus serpyllum, and Artemisia annua inhibit spike protein of SARS-CoV-2 binding to ACE2 receptor: in silico approach. Curr Pharmacol Rep. https://doi.org/10.1007/s40495-021-00259-4
Rosmalena R, Elya B, Dewi BE, Fithriyah F, Desti H, Angelina M, Hanafi M, Lotulung PD, Prasasty VD, Seto D (2019) The antiviral effect of indonesian medicinal plant extracts against dengue virus in vitro and in silico. Pathogens 8(2):85. https://doi.org/10.3390/pathogens8020085
Shukla R, Tripathi T (2020) Molecular dynamics simulation of protein and protein–ligand complexes. Computer-aided drug design. Springer, Singapore, pp 133–161. https://doi.org/10.1007/978-981-15-6815-2_7.
Trujillo-Correa AI, Quintero-Gil DC, Diaz-Castillo F, Quiñones W, Robledo SM, Martinez-Gutierrez M (2019) In vitro and in silico anti-dengue activity of compounds obtained from Psidium guajava through bioprospecting. BMC Complement Alternative Med. https://doi.org/10.1186/s12906-019-2695-1
Author Information
Department of Chemistry & Biochemistry, School of Basic Sciences and Research, Sharda University, Greater Noida, India