An efficient genomic DNA isolation protocol from juvenile leaves of Toona ciliata M. Roem
*Article not assigned to an issue yet
Neha, Meena Rajendra K., Bhandari Maneesh S., Garg Rimpee, Yadav Abhishek, Sharma Vipul, Kant Rama
Research Articles | Published: 22 February, 2026
First Page: 0
Last Page: 0
Views: 593
Keywords: Genomic DNA, Secondary metabolites, Polymerase chain reaction, Cetyl trimethylammonium bromide (CTAB), Leaf tissues
Abstract
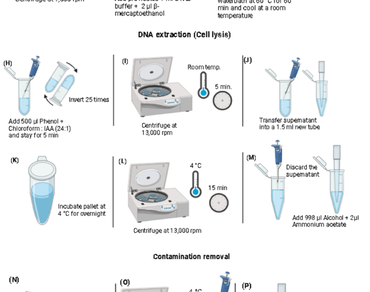
Consistent measurement of DNA concentration and purity is vital for all molecular studies. In the current extraction protocol, 0.5 g of juvenile leaf tissues were used to check the efficiency of DNA extraction. The quality and quantity of genomic DNA were tested on 0.8% agarose gel and a UV-visible spectrophotometer. The suitability of extracted genomic DNA for its use in molecular marker assays, particularly Simple Sequence Repeats. The DNA yield ranged from 306 to 1693 ng/µL, with an absorbance ratio between 1.75−1.92, respectively. In this protocol, elevated concentrations of CTAB, PVP, and β-mercaptoethanol were used to effectively remove proteins, tannins, and polyphenols. The effective PCR amplification of the leaf tissues confirmed that the genomic DNA was of good quality at the initial concentration of 40ng/ µL. Thus, the modified CTAB method (M2) can therefore be employed to extract high-quality genomic DNA from juvenile leaf tissues of T. ciliata, which predominantly contain proteins and tannins.
References
Amani J, Kazemi R, Abbasi AR, Salmanian AH (2011) A simple and rapid leaf genomic DNA extraction method for polymerase chain reaction analysis. Iran J Biotechnol 9(1):69–71
Atkinson BA (2020) Fossil evidence for a cretaceous rise of the Mahogany family. Am J Bot 107(1):139–147. https://doi.org/10.1002/ajb2.1416
Brunt C, Read J, Sanson GD (2006) Changes in resource concentration and defence during leaf development in a tough-leaved (Nothofagus moorei) and soft-leaved (Toona ciliata) species. Oecologia 148:583–592. https://doi.org/10.1007/s00442-006-0369-4
Carrier G, Santoni S, Rodier- Goud M, Canaguier A, De Kochko A, Dubreuil- Tranchant C, This P, Boursiquot J-M, Le Cunff L (2011) An efficient and rapid protocol for plant nuclear DNA Preparation suitable for next generation sequencing methods. Am J Bot 98:13–15. https://doi.org/10.3732/ajb.1000371
Chang JY (1997) A two-stage mechanism for the reductive unfolding of disulfide-containing proteins. J Biol Chem 272(1):69–75
Doyle J, Doyle J (1987) A rapid procedure for DNA purification from small quantities of fresh leaf tissues. Phytochem Bull 19:11–15
Ginwal HS, Maurya SS (2010) Evaluation and optimization of DNA extraction method for Dalbergia Sissoo leaf. Indian J Biotechnol 91(1):69–73
Horne EC, Kumpatla SP, Patterson KA, Gupta M, Thompson SA (2004) Improved high-throughput sunflower and cotton genomic DNA extraction and PCR fidelity. Plant Mol Bio Rep 22:83–84. https://doi.org/10.1007/BF02773352
Kenzo T, Yoneda R, Tanaka-Oda A, Azani MA (2018) Growth performance and leaf ecophysiological traits in three Aquilaria species in Malaysia. New for 50:699–715. https://doi.org/10.1007/s11056-018-09693-7
Koenen EJM, Clarkson JJ, Pennington TD, Chatrou IW (2015) Recently evolved diversity and convergent radiations of rainforest Mahogany (Meliaceae) shed new light on the origins of rainforest hyperdiversity. New Phytol 207:327–339. https://doi.org/10.1111/nph.13490
Li JT, Yang J, Chen DC, Zhang XL, Tang ZS (2007) An optimized mini-preparation method to obtain high-quality genomic DNA from mature leaves of sunflower. Genet Mol Res 6(4):1064–1071
Michiels A, Van den Ende W, Tucker M, Van Riet L LaereA (2003) Extraction of high-quality genomic DNA from latex-containing plants. Anal Biochem 315(1):85–89. https://doi.org/10.1016/s0003-2697(02)00665-6
Murray MG, Thompson W (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8(19):4321–4326. https://doi.org/10.1093/nar/8.19.4321
Neha, Kant R, Bhandari MS, Meena RK, Shankhwar R (2024) Genetic diversity and population structure in natural populations of Toona ciliata in the Uttarakhand state of himalaya. Trees 38:357–372. https://doi.org/10.1007/s00468-023-02487-1
Porebski S, Bailey LG, Baurn BR (1997) Modification of a CTAB DNA extraction protocol for plants containing high polysaccharide and polyphenol components. Plant Mol Bio Rep 15:8–15. https://doi.org/10.1007/BF02772108
Rajarajan K, Devi SV, Handa AK, Gurunathan N, Dhyani SK, Uthappa AR (2015) A simple and effective method for high quality PCR usable DNA extraction from Azadirachta indica A. Juss. IJA 17(1):24–28
Rawat S, Joshi G, Annapurna D, Arunkumar A, Karaba N (2016) Standardization of DNA extraction method from mature dried leaves and ISSR-PCR conditions for Melia dubia Cav. –A fast growing multipurpose tree species. Am J Plant Sci 7:437–445. https://doi.org/10.4236/ajps.2016.73037
Sahu SK, Thangraj M, Kathiresen K (2012) DNA extraction protocol for plants with high levels of secondary metabolites and polysaccharides without using liquid nitrogen and phenol. Mol Bio 2012:1–6. https://doi.org/10.5402/2012/205049
Sangwan NS, Sangwan RS, Kumar S (1998) Isolation of genomic DNA from the antimalarial plant Artemisia annua. Plant Mol Bio Rep 16:365–365
Semagn K (2014) Leaf tissue sampling and DNA extraction protocols. Molecular plant taxonomy, vol 1115. Humana, Totowa, NJ, USA, pp 53–67. https://doi.org/10.1007/978-1-62703-767-9_3
Sharma S, Kumar A, Dobhal S, Rawat A (2018) Standardization of protocol for genomic DNA isolation in Melia compositaBenth.(SM dubia Cav). Indian J Biotechnol 17:185–188
Syamkumar S, Lowarence B, Sasikumar B (2003) Isolation and amplification of DNA from rhizomes of turmeric and ginger. Plant Mol Biol Rep 21:171a-171e https://doi.org/10.1007/BF02774243
Varma A, Padh H, Shrivastava N (2007) Plant genomic DNA isolation: an Art or a science. Biotechnol Healthc 2(3):386–392. https://doi.org/10.1002/biot.200600195
Williams CE, Ronald PC (1994) PCR template-DNA isolated quickly from monocot and Dicot leaves without tissue homogenization. Nucleic Acids Res 22(10):1917–1918. https://doi.org/10.1093/nar/22.10.1917
Sarwat M, Negi MS, Lakshmikumaran M, Tyagi AK, Das S, Srivastava PS (2006) A standardized protocol for genomic DNA isolation from Terminalia Arjuna for genetic diversity analysis. Electron J Biotechnol 9(1):86–91
Puchooa D, Khoyratty SUSS (2004) Genomic DNA extraction from Victoria Amazonica. Plant Mol Bio Rep 22(2):195a–195i
Author Information
Division of Genetics & Tree Improvement, ICFRE-Forest Research Institute, Dehradun, India