Phytochemical, enzymatic antioxidant, and nonenzymatic antioxidant metabolism during germination of Cajanus scarabaeoides seeds
*Article not assigned to an issue yet
Rokkam Rajesh, Pinipay Felicity, Botcha Satyanarayana, Tamanam Raghava Rao
Research Articles | Published: 25 April, 2024
Online ISSN : 2229-4473.
Website:www.vegetosindia.org
Pub Email: contact@vegetosindia.org
First Page: 0
Last Page: 0
Views: 618
Keywords:
n Cajanus scarabaeoidesn , Germination, Antioxidants, Phytochemicals, Seeds
Abstract
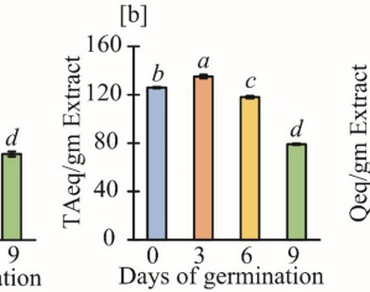
Key stages of the plant life cycle like germination and seedling growth are highly impacted by physical and biochemical parameters. This study aims to investigate the germination and seedling growth and development of Cajanus scarabaeoides which is widespread in the Asian region and growing spontaneously in India. During 0, 3, 6, and 9th day of germination of C. scarabaeoides seeds, phytochemical components like total phenolic content (TPC), total tannin contents (TTC), total flavonoid contents (TFC), enzymatic antioxidants like superoxide dismutase (SOD), catalase (CAT), peroxidase (POD) in correlation with protein concentrations and non-enzymatic antioxidants like 2,2-diphenyl-1-picrylhydrazyl (DPPH), Ferric Reducing Antioxidant Power (FRAP), and Reducing power activity were measured. The results showed augmented levels of phytochemicals on the 0th day and likewise for proteins and enzymatic oxidants on the 9th day. However, for non-enzymatic antioxidants, high DPPH activity was found on the 9th day, FRAP on the 0th day, and reducing power on the 3rd day of germination. Therefore, this study demonstrates how the antioxidant system is effectively activated and the synergistic activities of antioxidants compared to phytochemicals to support C. scarabaeoides seed germination. To use C. scarabaeoides seed in the creation of functional food, germination can be a useful technique to boost the nutritional and nutraceutical potential of the seed.
(*Only SPR Members can get full access. Click Here to Apply and get access)
References
Abdul-Baki AA, Anderson JD (1973) Vigor determination in soybean seed by multiple criteria 1. Crop Sci 13(6):630–633. https://doi.org/10.2135/cropsci1973.0011183X001300060013x
Ali AS, Elozeiri AA (2017) Metabolic processes during seed germination. Adv Seed Biol 2017:141–166. https://doi.org/10.5772/intechopen.70653
Al-Mudaris M (1998) Notes on various parameters recording the speed of seed germination. Der Tropenlandwirt 99:147–154
Anand A et al (2019) Hydrogen peroxide signaling integrates with phytohormones during the germination of magnetoprimed tomato seeds. Sci Rep 9(1):8814. https://doi.org/10.1038/s41598-019-45102-5
Bao J, Cai Y, Sun M, Wang G, Corke H (2005) Anthocyanins, flavonols, and free radical scavenging activity of Chinese bayberry (Myrica rubra) extracts and their color properties and stability. J Agric Food Chem 53(6):2327–2332. https://doi.org/10.1021/jf048312z
Beauchamp BC, Fedovich I (1976) Superoxide dismutase: improved assay and an assay applicable to acrylamide gel. Anal Biochem 10:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Benzie IFF, Strain JJ (1996) The Ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Bewley JD et al (2013) Germination. Seeds: physiology of development, germination and dormancy, 3rd edn. Springer, New York, pp 133–181. https://doi.org/10.1007/978-1-4614-4693-4_4
China Saxena KB, Zhou C, Zhang C, Li Z, Zhang J, Liu X (2002) Characterization of Cajanus scarabaeoides growing in Yuanjiang county of Yunnan province in China. International Chickpea and Pigeonpea Newsletter, 9. pp 34–37. http://oar.icrisat.org/id/eprint/4016. Accessed 10 Dec 2022
Cuendet M, Hostettmann K, Potterat O (1997) Iridoidglucosides with free scavenging properties from Fagracablumei. Helv Chim Acta 80:1144–1152. https://doi.org/10.1002/hlca.19970800411
Dutta A et al (2022) A comprehensive review on grain legumes as climate-smart crops: challenges and prospects. Environ Chall. https://doi.org/10.1016/j.envc.2022.100479
Folin O, Ciocalteu V (1927) On tyrosine and tryptophane determinations in proteins. J Biol Chem 73(2):627–650. https://doi.org/10.1016/S0021-9258(18)84277-6
Fowler JL (1991) Interaction of salinity and temperature on the germination of crambe. Agron J 83(1):169–172. https://doi.org/10.2134/agronj1991.00021962008300010039x
Foyer CH, Noctor G (2005) Oxidant and antioxidant signalling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ 28(8):1056–1071. https://doi.org/10.1111/j.1365-3040.2005.01327.x
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48(12):909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Grygier A (2021) Mustard seeds as a bioactive component of food. Food Rev Int. https://doi.org/10.1080/87559129.2021.2015774
Javanmardi J, Stushnoff C, Locke E, Vivanco JM (2003) Antioxidant activity and total phenolic content of Iranian Ocimum accessions. Food Chem 83(4):547–550. https://doi.org/10.1016/S0308-8146(03)00151-1
Jolliffe IT, Cadima J (2016) Principal component analysis: a review and recent developments. Phil Trans R Soc A 374(2065):20150202. https://doi.org/10.1098/rsta.2015.0202
Kasote DM et al (2015) Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int J Biol Sci 11(8):982. https://doi.org/10.7150/ijbs.12096
Kirtikar K R, Basu BD (1975) Indian Medicinal Plants, 2nd ed, Vol. III, Dehra Dun, Lalit Mohan Basu, Allahabad, pp 2327–2328
Kumari S, Krishnan V, Sachdev A (2015) Impact of soaking and germination durations on antioxidants and anti-nutrients of black and yellow soybean (Glycine max. L) varieties. J Plant Biochem Biotechnol 24:355–358. https://doi.org/10.1007/s13562-014-0282-6
Laxmi G, Chaturvedi N, Richa S (2015) The impact of malting on nutritional composition of foxtail millet, wheat and chickpea. J Nutr Food Sci. https://doi.org/10.4172/2155-9600.1000407
Li Z, Lu W, Yang L, Kong X, Deng X (2015) Seed weight and germination behavior of the submerged plant Potamogeton pectinatus in the arid zone of northwest China. Ecol Evol 5:1504–1512. https://doi.org/10.1002/ece3.1451
Li W et al (2022) Advances in the understanding of reactive oxygen species-dependent regulation on seed dormancy, germination, and deterioration in crops. Front Plant Sci. https://doi.org/10.3389/2Ffpls.2022.826809
Lopez-Amoros ML, Hernandez T, Estrella I (2006) Effect of germination on legume phenolic compounds and their antioxidant activity. J Food Comp Anal 19:277–283. https://doi.org/10.1016/j.jfca.2004.06.012
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Lozano-Isla F, Benites-Alfaro OE, Pompelli MF (2019) GerminaR: an R package for germination analysis with the interactive web application “GerminaQuant for R.” Ecol Res 34(2):339–346. https://doi.org/10.1111/1440-1703.1275
Lv X, Meng G, Li W, Fan D, Wang X, Espinoza-Pinochet CA, Cespedes-Acuña CL (2020) Sulforaphane and its antioxidative effects in broccoli seeds and sprouts of different cultivars. Food Chem 316:126216. https://doi.org/10.1016/j.foodchem.2020.126216
Mandal SC, Maity TK, Das J, Saha BP, Pal M (2000) Anti- inflammatory evaluation of ficus racemosa leaf extract. J Ethnopharmacol 72(1–2):87–92
Moongngarm A, Saetung N (2010) Comparison of chemical compositions and bioactive compounds of germinated rough rice and brown rice. Food Chem 122(3):782–788. https://doi.org/10.1016/j.foodchem.2010.03.053
Nonogaki H, Bassel GW, Derek Bewley J (2010) Germination—still a mystery. Plant Sci 179(6):574–581. https://doi.org/10.1016/j.plantsci.2010.02.010
Orchard TJ (1977) Estimating the parameters of plant seedling emergence. Seed Sci Technol 5:61–69
Peduruhewa PS, Jayathunge KGLR, Liyanage R (2021) Potential of underutilized wild edible plants as the food for the future–a review. J Food Secur 9(4):136–147. https://doi.org/10.12691/jfs-9-4-1
Quisumbing E (1978) Medicinal Plants of the Phillippines 1978 Katha Publishing Co. Inc. Phil 977. https://www.cabdirect.org/cabdirect/abstract/20057007481. Accessed 12 Dec 2022
Radhakrishnan TM, Sarma PS (1963) Intracellular localization and biosynthesis of catalase in liver tissues. Curr Sci 32:1749
Ranal MA, Santana DG (2006) How and why to measure the germination process? Braz J Bot 29(1):1–1. https://doi.org/10.1590/S0100-84042006000100002
Ray AS, Rahaman CH (2018) Pharmacognostic standardization and phytochemical investigation of Cajanus scarabaeoides (L.) Thouars. Res J Pharmacogn Phytochem 10(1):120–131. https://doi.org/10.5958/0975-4385.2018.00018.3
Ray AS et al (2018) Polyphenol enriched ethanolic extract of Cajanus scarabaeoides (L.) Thouars exerts potential antifilarial activity by inducing oxidative stress and programmed cell death. PLoS ONE 13(12):e0208201. https://doi.org/10.1371/journal.pone.0208201
Sadasivam S, Manickam A (1996) Biochemical methods, 2nd edn. New Age International Ltd Publishers, New Delhi, pp 108–110
Scott SJ, Jones RA, Williams WA (1984) Review of data analysis methods for seed germination. Crop Sci 24(6):1192–1199. https://doi.org/10.2135/cropsci1984.0011183X002400060043x
Siddiqui AA, Ali M (1997) Practical pharmaceutical chemistry. CBS Publishers & Distributors. https://doi.org/10.12691/ajbr-3-1-2
Stagnari F et al (2017) Multiple benefits of legumes for agriculture sustainability: an overview. Chem Biol Technol Agric 4(1):1–13. https://doi.org/10.1186/s40538-016-0085-1
Upadhyaya H, Reddy K, Sube S, Gowda L, Cholenahalli. (2012) Phenotypic diversity in Cajanus species and identification of promising sources for agronomic traits and seed protein content. Genetic Resour Crop Evolut. https://doi.org/10.1007/s10722-012-9864-0.10.1007/s10722-012-9864-0
Zhao M et al (2018) Mobilization and role of starch, protein, and fat reserves during seed germination of six wild grassland species. Front Plant Sci 9:234. https://doi.org/10.3389/fpls.2018.00234
Zhou W, Chen F, Luo X, Dai Y, Yang Y, Zheng C, Shu K (2020) A matter of life and death: molecular, physiological, and environmental regulation of seed longevity. Plant, Cell Environ 43(2):293–302. https://doi.org/10.1111/pce.13666
Acknowledgements
Author Information
College of Science and Technology, Andhra University, Visakhapatnam, India
rrajesh125@gmail.com
College of Science and Technology, Andhra University, Visakhapatnam, India
felicityosteen@gmail.com
College of Science and Technology, Andhra University, Visakhapatnam, India
satyabiochemau@gmail.com
College of Science and Technology, Andhra University, Visakhapatnam, India
trrao_au@yahoo.com