In vivo study of efficacy of synthesized silver nanoparticle for quercetin from Bacillus Subtilis against diabetic mice model
Research Articles | Published: 04 September, 2022
Online ISSN : 2229-4473.
Website:www.vegetosindia.org
Pub Email: contact@vegetosindia.org
First Page: 93
Last Page: 105
Views: 1462
Keywords:
Quercetin,
Bacillus subtilis
, Nanoparticle, Wound healing, Diabetic mice
Abstract
Quercetin has significant therapeutic applications such as anticancer, anti-fibrosis, and anti-inflammatory agent. It is mainly found in different parts of plant, but, major challenge is the involvement of costly downstream processing techniques for its biosepration. Therefore, in this study, microbial fermentative approach has been employed to achieve its large production at very low cost. Bacillus subtilis MTCC1305 was selected as potent strain for producing intracellular quercetin with production yield of 108.08 mg/ml. The production was enhanced up to 178.58 mg/ml after optimization of media components and fermentation conditions. Development of silver nanoparticle for this purified extract of quercetin has overcame their limitation of poor bioavailability within body fluid. Scanning electron microscopy (SEM) had confirmed the uniform size of its silver nanoparticle as 260 nm. The encapsulation efficiency was obtained as 58% for this silver impregnated purified quercetin extract. The synthesis of nanoparticle was confirmed by Zeta potential value as − 39.5 kV with single peak without any noise property. In diabetic mice model, the nanoparticle treated mice showed significant wound healing during 14th day of treatment. In these treated diabetic mice model, the reepithelization was observed with appearance of different layers of skin similar to that of the control mice. These mice showed improved antioxidant properties with increased level of superoxide dismutase (SOD, 3.6%), catalase (CAT-7.5%), glutathione peroxidase (GPx-4.5%), reduced form of glutathione (GSH-2%) and lipid peroxidase (LPO-10%). These factors showed their effective role for delayed wound healing in diabetic mice.
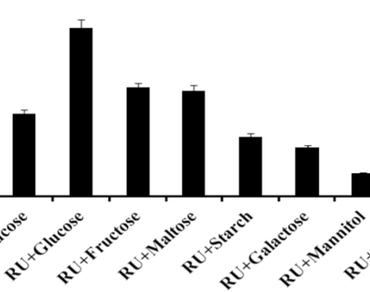
(*Only SPR Members can get full access. Click Here to Apply and get access)
References
Aebi H (1984) Catalase in vitro. Method Enzyme 105:121–126
Barua CC, Talukdar A, Begum SA, Buragohain B, Roy JD, Pathak DC, Sarma DK, Gupta AK, Bora RS (2012) Effect of Alternanthera brasiliana (L) Kuntze on healing of dermal burn wound. Indian J Exp Biol 50:56–60
Chitkara D, Nikalaje SK, Mittal A, Chand M, Kumar N (2012) Development of quercetin nanoformulation and in vivo evaluation using streptozotocin induced diabetic rat model. Drug Deliv Transl Res 2(2):112–123
Cohn VH, Lyle J (1966) Fluorimetric assay for glutathione. Analyticalbiochemistry 14(3):434–440
Doersch KM, Newell-Rogers MK (2017) The impact of quercetin on wound healing relates to changes in αV and β1 integrin expression. Exp Biol Med 242(14):1424–1431
Dogan E, Yanmaz L, Gedikli S, Ersoz U, Okumus Z (2017) The effect of pycnogenol on wound healing in diabetic rats. Ostomy Wound Manage 63(4):41–47
Flohe L, Gunzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105(1):114–121
Fu J, Huang J, Lin M, Xie T, You T (2020) Quercetin promotes diabetic wound healing via switching macrophages from M1 to M2 polarization. J Surg Res 246:213–223
Gomathi K, Gopinath D, Rafiuddin Ahmed M, Jayakumar R (2003) Quercetin incorporated collagen matrices for dermal wound healing processes in rat. Biomaterials 24(2003):2767–2772
Gonzalez AL, Cecilia Noguez J, Beranek AS (2014) Size, shape, stability, and color of plasmonic silver nanoparticles. J Phys Chem C 118:9128–9136
Gupta R, MathurM VKB, Katariya P, Yadav S, Kamal R, Gupta RS (2012) Evaluation of antidiabetic and antioxidant activity of Moringa oleifera in experimental diabetes. J Diabetes 4:164–171
Hamdan S, Pastar I, Drakulich S, Dikici E, Tomic-Canic M, Deo S et al (2017) Nanotechnology-driven therapeutic interventions in wound healing: potential uses and applications. ACS Cent Sci 3:163–175b
Han X, Tao Y, Deng Y, Yu J, Sun Y, Jiang G (2017) Metformin accelerates wound healing in type 2 diabetic db/db mice. Mol Med Rep 16(6):8691–8698
Hay GW, Westlake DWS, Simpson FJ (1961) Degradation of rutin by Aspergillus flavus: purification and characterization of rutinase. Can J Microbiol 7:921–932
Hendson M, Hildebrand DC, Schroth MN (1992) Distribution among Pseudomonads of sequences homologous to the rutin glycosidase and P-glucosidase genes of Pseudomonas viridiflava. Phytopathology 82:1230–1233
Jangde R, Srivastava S, Singh MR, Singh D (2018) In vitro and In vivo characterization of quercetin loaded multiphase hydrogel for wound healing application. Int J Biol Macromol 115:1211–1217
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys 21:130–132
Kavitha C, Indira G (2016) Quantitative estimation of total phenolic, flavonoids, tannin and chlorophyll content of leaves of StrobilanthesKunthiana (Neelakurinji). J Med Plants 4:282–286
Kim YS, Cho IH, Jeong MJ, Jeong SJ, Nah SY, Cho YS, Kim SH, Go A, Kim SE, Kang SS, Moon CJ, Kim JC, Kim SH, Bae CS (2011) Therapeutic effect of total ginseng saponin on skin wound healing. J Ginseng Res 35:360–367
Nam HK, Hong SH, Shin KC, Oh DK (2012) Quercetin production from rutin by a thermostable β-rutinosidase from Pyrococcusfuriosus. Biotechnol Lett 34:483–489
Narikawa T, Shinoyama H, Fujii T (2000) A beta-rutinosidase from Penicilliumrugulosum IFO 7242 that is a peculiar flavonoid glycosidase. Biosci Biotechnol Biochem 64:1317–1319
Negm NA, Tawfik SM, Abd-Elaal AA (2015) Synthesis, characterization and biological activity of colloidal silver nanoparticles stabilized by gemini anionic surfactants. J Ind Eng Chem 21:1051–1057
Polerà N, Badolato M, Perri F, Carullo G, Aiello F (2019) Quercetin and its natural sources in wound healing management. Curr Med Chem 26(31):5825–5848
Pompella A, Visvikis A, Paolicchi A, De Tata V, Casini AF (2003) The changing faces of glutathione, a cellular protagonist. Biochem Pharmacol 66(8):1499–1503
Ponrasu T, Kannappan MS, Ganeshkumar M, Suguna L (2013) Role of Annona squamosa on antioxidants during wound healing in streptozotocin nicotinamide induced diabetic rats. J PharmacogPhytochem 2(4):77–84
Sahu N, Soni D, Chandrashekhar B, Satpute DB, Saravanadevi S, Sarangi BK, Pandey RA (2016) Synthesis of silver nanoparticles using flavonoids: hesperidin, naringin and diosmin, and their antibacterial effects and cytotoxicity. International Nano Letters. https://doi.org/10.1007/s40089-016-0184-9
Seguí J, Gironella M, Sans M, Granell S, Gil F, Gimeno M, Coronel P, Piqué JM, Panés J (2004) Superoxide dismutase ameliorates TNBS- induced colitis by reducing oxidative stress, adhesion molecule expression, and leukocyte recruitment into the inflamed intestine. J Leukoc Biol 76(3):537–544
Shafiq-Ur-Rehman (1984) Lead-induced regional lipid peroxidation inbrain. Toxicol Lett 21(3):333–337
Striccoli M, Sandra MB et al (2012) Antioxidant effects of quercetin and catechin encapsulated into PLGA nanoparticles. J Nanomater 2012:1–12
Shirwaikar A, Somashekar AP, Udupa AL, Udupa SL, Somashekar S (2003) Wound healing studies of the Aristolochia bracteolate Lam with supportive action of antioxidant enzymes. Phytomedicine 10:558–562
Shrivastava S, Bera T, Singh SK, Singh G, Ramachandrarao P, Dash D (2009) Characterization of antiplatelet properties of silver nanoparticles. ACS Nano 3:1357–1364
Stensberg MC, Wei Q, McLamore ES, Porterfield DM, Wei A, Sepúlveda MS (2011) Toxicological studies on silver nanoparticles: challenges and opportunities in assessment, monitoring and imaging. Nanomedicine 6(5):879–898
Sung JH, Ji JH, Park JD, Yoon JU, Kim DS, Jeon KS, Song MY, Beom SH, Jeong HH, Yong HC, Hee KC, Ji HL, Myung HC, Bruce JK, Il JYu (2009) Subchronic inhalation toxicity of silver nanoparticles. Toxicol Sci 108(2):452–461
Tasca F, Antiochia R (2020) Biocide activity of green quercetin-mediated synthesized silver nanoparticles. Nanomaterials 10(5):909. https://doi.org/10.3390/nano10050909
Venukumar MR, Latha MS (2002) Antioxidant activity of Curculigoorchioides in carbon tetrachloride-induced hepatopathy in rats. Indian J Clin Biochem 17:80–87
Wach A, Pyrzynska K, Biesaga M (2007) Quercetin content in some food and herbal samples. Food Chem 100(2):699–704
Wang J, Zhao LL, Sun GX, Liang Y, Wu FA, Chen ZL, Cui SM (2011) A comparison of acidic and enzymatic hydrolysis of rutin. Afr J Biotech 10:1460–1466
Wang YD, Chen WD, Li C, Guo C, Li Y, Qi H, Shen H, Kong J, Long X, Yuan F, Wang X, Huang W (2015) Farnesoid X receptor antagonizes JNK signaling pathway in liver carcinogenesis by activating SOD3. Mol Endocrinol 29:322–331
Acknowledgements
Author Information
Institute of Allied Medical Science and Technology, NIET, NIMS University, Rajasthan, India
priyay20@gmail.com