Optimization of production parameter and application of glycoprotein produced from Sphingobacterium thalpophilum DP9
Research Articles | Published: 28 September, 2021
Online ISSN : 2229-4473.
Website:www.vegetosindia.org
Pub Email: contact@vegetosindia.org
First Page: 26
Last Page: 37
Views: 1797
Keywords:
Biosurfactant, Central Composite Design, Cosmetics, Glycoprotein, Shampoo
Abstract
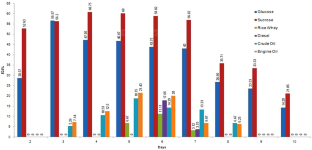
Biosurfactants are biologically secreted amphiphilic molecules which resemble with the chemical surfactant in function. Compared to the chemical surfactants, they are biodegradable, biocompatible and less toxic. However, commercialization of biosurfactant production is still challenging due to high production and recovery cost, less efficient strain availability, and less knowledge about biosynthetic or metabolic pathways of biosurfactant production. Therefore, the attempt was made to optimize the production parameter for improving the biosurfactant production using Central Composite Design (CCD) from Sphingobacterium thalpophilum DP9. In addition, application of biosurfactant as shampoo ingredient was also studied. Results showed that the biosurfactant production was increased by 4.94 fold when 1% bacterial inoculum was inoculated in Bushnell-Hass medium (pH–11) supplemented with 8 g% sucrose. Biosurfactant was effectively purified by acid precipitation from cell free supernatant. Chromatographic techniques (TLC and HPTLC) and Native–PAGE electrophoresis confirmed glycoprotein nature of the biosurfactant with 85.75 ± 0.05% carbohydrate and 4.58 ± 0.03% protein part. In addition, two fragments of protein (37 kD and 44 kD) were also observed in electrophoretic analysis. According to the ‘blind touch test’ data of volunteer it was noted that the biosurfactant based shampoo showed satisfactory (conditioning) activity compared to commercially available shampoo. Therefore, it was concluded that glycoprotein produced from S. thalpophilum DP9 can be used in cosmetic or personal health products.
(*Only SPR Members can get full access. Click Here to Apply and get access)
References
According to web reference [https://newsweeekgroup.com/2019/06/25/biosurfactants-market-trends-2019-emerging-technologies-sales-revenue-key-players-analysis-development-status-opportunity-assessment-and-industry-expansion-strategies-2022/]
Al-Dhabi NA, Esmail GA, Arasu MV (2020) Enhanced production of bosirfactant from Bacillus subtilis strain Al-Dhabi-130 under solid-state fermentation using date molasses from Saudi Arabia for bioremediation of cruid oi contaminated soils. Int J Environ Res Public Health 17:8446. https://doi.org/10.3390/ijerph17228446
Amani H, Haghighi M, Keshtkar MJ (2013) Production and optimization of microbial surfactin by Bacillus subtilis for ex situ enhanced oil recovery. Petrol Sci Technol 31:1249–1258. https://doi.org/10.1080/10916466.2010.542416
Arndt C, Koristka S, Bartsch H, Bachmann M (2012) Native Polyacrylamide Gels. In: Scofield BTKH (ed) Protein electrophoresis: methods and protocols, methods in molecular biology. Humana Press, Totowa, NJ, pp 49–53
Badi K, Khan S (2014) Formulation, evaluation and comparision of herbal shampoo with commercial shampoo. Beni-Suef University J Basic Appl Sci 3:301–305. https://doi.org/10.1016/j.bjbas.2014.11.005
Bakhshi N, Sheikh-Zeinoddin M, Soleimanian-Zad S (2018) Production and partial characterization of a glycoprotein bioemulsifier produced by Lactobacillus plantarum subsp. plantarum PTCC 1896. J Agric Sci Technol 20(1):37–49
Bazsefidpar S, Mokhtarani B, Panahi R, Hajfarajollah H (2019) Overproduction of rhamnolipid by fed-batch cultivation of Pseudomonas aeruginosa in a lab-scale fermenter under tight DO control. Biodegradation 30:59–69. https://doi.org/10.1007/s10532-018-09866-3
Bezerra GO, Rufino RD, Luna JM, Sarubbo LA (2018) Saponins and microbial biosurfactants: potential raw materials for the formulation of cosmetics. Biotechnol Progress 34:1482–1493. https://doi.org/10.1002/btpr.2682
Bezerra H, Sobrinho DS, Moura J et al (2013) Assessment of toxicity of a biosurfactant from Candida sphaerica UCP 0995 cultivated with industrial residues in a bioreactor. Electron J Biotechnol 16:1–12. https://doi.org/10.2225/vol16-issue4-fulltext-4
Brown M (1991) Biosurfactants for cosmetic applications. Int J Cosmetic Sci 13:61–64
Brumano LP, Antunes FAFA, Souto SG, Santos JC, Schneider R, Silverio da Silva S (2017) Biosurfactant production by Aurobasidium pullulans in stirred tank bioreactor: new approch to understand the influence of important variables in the process. Biores Technol 243:264–272. https://doi.org/10.1016/j.biortech.2017.06.088
Chavan VM, Tiwari KJ, Suryavanshi KA, Bhor AS (2019) Formulation and evaluation of herbal shampoo. Am J PharmTech Res 9(5):88–96
Fadhile A, Abu H, Idris M et al (2016) Biosurfactant production by the hydrocarbon-degrading bacteria ( HDB ) Serratia marcescens : optimization using central composite design ( CCD ). J Indust Eng Chem 47:272–280. https://doi.org/10.1016/j.jiec.2016.11.043
Golhani D, Pandey V, Shukla A, Shukla R (2015) Formulation and comparative evalation of herbal shampoo with marketed products. Mintage J Pharma Med Sci 4:4–7
Gour VS, Sanadhya N, Sharma P et al (2015) Biosurfactant characterization and its potential to remove sebum from hair. Indust Crops and Products 69:462–465. https://doi.org/10.1016/j.indcrop.2015.03.007
Hogan DE, Tian F, Malm SW et al (2019) Biodegradability and toxicity of monorhamnolipid biosurfactant diastereomers. J Hazard Material 364:600–607. https://doi.org/10.1016/j.jhazmat.2018.10.050
Joshi SJ, Desai AJ (2013) Bench-scale production of biosurfactants and their potential in ex-situ MEOR application. Soil Sediment Contam 22:701–715. https://doi.org/10.1080/15320383.2013.756450
Joy S, Rahman PKSM, Khare SK, Sharma S (2019) Production and characterization of glycolipid biosurfactant from Achromobacter sp. (PS1) isolate using one –factor–at –a–time (OFAT) approach with feasible utilization of ammonia–soaked lignocellulosic pretreated residues. Biopro Biosys Eng. https://doi.org/10.1007/s00449-019-02128-3
Kanlayavattanakul M, Lourith N (2010) Lipopeptides in cosmetics. Int J Cosmetic Sci 32:1–8. https://doi.org/10.1111/j.1468-2494.2009.00543.x
Kumar R, Das AJ (2017) Application of rhamnolipids in agriculture and food industry. In: Rhamnolipid biosurfactant. Springer Nature Singapore Pte Ltd. 2018, Singapore, pp 97–107. https://doi.org/10.1007/978-981-13-1289-2_9
Liu X, Ren B, Gao H, Liu M, Dai H, Song F, Yu Z, Wang S, Hu J, Kokare C, Zhan L (2012) Optimization for production of surfactin with new synergistic antifungal activity. PLoS ONE 7(5):e34430. https://doi.org/10.1371/journal.pone.0034430
Lourith N, Kanlayavattanakul M (2009) Natural surfactants used in cosmetics: glycolipids. Int J Cosmetic Sci 31:255–261. https://doi.org/10.1111/j.1468-2494.2009.00493.x
Lowry OH, Roserbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin-phenol reagent. J Biol Chem 193:262–267
Lukic M, Pantelic I, Savic S (2016) An overview of novel surfactants for formulation of cosmetics with certain emphasis on acidic active substances. Tenside Surf Deterg 53:7–19. https://doi.org/10.3139/113.110405
Makkar RS, Technology M, Increased I et al (1997) Biosurfactant production by a thermophilic Bacillus subtilis strain. J Ind Microbiol Biotechnol 18:37–42
Malviya R, Sharma PK (2014) Evaluation of marketed shampoo (synthetic and natural) for their hair cleansing, dirt dispersion, wetting time, solid content and foaming capacity properties. Global J Pharmacol 8:490–493. https://doi.org/10.5829/idosi.gjp.2014.8.4.84230
Markande AR, Acharya SR, Nerurkar AS (2013) Physicochemical characterization of a thermostable glycoprotein bioemulsifier from Solibacillus silvestris AM1. Process Biochem. https://doi.org/10.1016/j.procbio.2013.08.017
Mendhekar SY, Tajane AS, Shitole PB (2017) Formulation and evaluation of polyherb shampoo ad compare with marketed shampoo. World J Pharma Pharmaceut Sci 6:1388–1397. https://doi.org/10.20959/wjpps201712-10644
Mnif I, Ghribi D (2015) High molecular weight bioemulsifiers, main properties and potential environmental and biomedical applications. World J Microbiol Biotechnol 31:691–706. https://doi.org/10.1007/s11274-015-1830-5
Nalini S, Parthasarathi R (2018) Optimization of rhamnolipid biosurfactant production from Serratia rubidaea SNAU02 under solid-state fermentation and its biocontrol efficacy against Fusarium wilt of eggplant. Annal Agrarian Sci 16:108–115. https://doi.org/10.1016/j.aasci.2017.11.002
Ostendorf A, Silva IA, Converti A, Sarubbo LA (2019) Production and formulation of a new low-cost biosurfactant to remediate oil- contaminated seawater. J Biotechnol 295:71–79. https://doi.org/10.1016/j.jbiotec.2019.01.025
Parthipan P, Sarankumar RK, Jaganathan A (2018) Biosurfactants produced by Bacillus subtilis A1 and Pseudomonas stutzeri NA3 reduce longevity and fecundity of Anopheles stephensi and show high toxicity against young instars. Env Sci Pollution Res 25:10471–10481. https://doi.org/10.1007/s11356-017-0105-0
Pereira JFB, Gudina EJ, Costa R, Vitorino R, Texieria JA, Coutinho JA, Rodrigues LR (2013) Optimization and characterizatio of biosurfactant production by Bacillus subtilis isolates towards microbial enhaced oil recovery applications. Fuel 111:259–268. https://doi.org/10.1016/j.fuel.2013.04.040
Plackett RL, Burman JP (1946) The design of optimum multifactorial experiments. Biometrika 33(4):305–325. https://doi.org/10.2307/2332195
Price NPJ, Ray KJ, Vermillion KE et al (2012) Structural characterization of novel sophorolipid biosurfactants from a newly identified species of Candida yeast. Carbohyd Res 348:33–41. https://doi.org/10.1016/j.carres.2011.07.016
Ramalingam V, Varunkumar K, Ravikumar V, Rajaram R (2019) Production and structure elucidation of anticancer potential surfactin from marine actinomycete Micromonospora marina. Process Biochem 78:169–177. https://doi.org/10.1016/j.procbio.2019.01.002
Rikalovic M, Vrvic M, Karadzic I (2015) Rhamnolipid biosurfactant from Pseudomonas aeruginosa: from discovery to application in contemporary technology. J Serbian Chem Soc 80:279–304. https://doi.org/10.2298/JSC140627096R
Ron EZ, Rosenberg E (2001) Natural roles of biosurfactants. Minireview Env Microbiol 3:22–236. https://doi.org/10.1046/j.1462-2920.2001.00190.x
Santos EF, Teixeira MFS, Converti A et al (2019) Production of a new lipoprotein biosurfactant by Streptomyces sp. DPUA1566 isolated from lichens collected in the Brazilian Amazon using agroindustry wastes. Biocatal Agri Biotechnol 17:142–150. https://doi.org/10.1016/j.bcab.2018.10.014
Sekar M, Aqeela H, Noordin M, Merican N (2016) Formulation and evaluation of herbal shampoo containing rambutan leaves extract. Int J Pharma Biosci 7:146–151
Senthil S, Mani P, Kumar CG, Jayalakshmi S (2019) Structural characterization and biological evaluation of Staphylosan (dimannooleate), a new glycolipid surfactant produced by a marine Staphylococcus saprophyticus SBPS-15. Enz Microbial Technol 120:1–7. https://doi.org/10.1016/j.enzmictec.2018.09.008
Sergio G, Perez-ameneiro M, Vecino X, Pastoriza-santos I (2017) Biogenic synthesis of metal nanoparticles using a biosurfactant extracted from corn and their antimicrobial properties. Nanomat Article 7:1–14. https://doi.org/10.3390/nano7060139
Shahaliyan F, Safahieh A, Abyar H (2015) Evaluation of emulsification index in marine bacteria Pseudomonas sp. and Bacillus sp. Arab J Sci Eng 40:1849–1854. https://doi.org/10.1007/s13369-015-1663-4
Souza KST, Gudiña EJ, Schwan RF et al (2017) Improvement of biosurfactant production by Wickerhamomyces anomalus CCMA 0358 and its potential application in bioremediation. J Hazard Material 346:152–158. https://doi.org/10.1016/j.jhazmat.2017.12.021
Templeton RH (2018) Reetha and shikakai as natural surfactants for cleaning of historic textiles. Int J Res Analyt Rev 5:2348–2350
Vecino X, Cruz JM, Moldes AB, Rodrigues LR (2017) Critical reviews in biotechnology biosurfactants in cosmetic formulations : trends and challenges. Criti Rev Biotechnol 37(7):911–923. https://doi.org/10.1080/07388551.2016.1269053
Vera ECS, de Azevedo PODS, Domínguez JM, de Souza Oliveira RP (2018) Optimization of biosurfactant and bacteriocin-like inhibitory substance (BLIS) production by Lactococcus lactis CECT-4434 from agroindustrial waste. Biochem Eng J 133:168–178. https://doi.org/10.1016/j.bej.2018.02.011
Acknowledgements
Authors are thankful to the Head of Department, Post Graduate Department of Biosciences and Sardar Patel University (Vallabh vidyanagar, Anand, Gujarat, India) for providing the platform to carry out this research work.
Author Information
Post Graduate Department of Bioscience, Sardar Patel University, Anand, India
dhaval30103@gmail.com
Post Graduate Department of Bioscience, Sardar Patel University, Anand, India
jytsolanky269@gmail.com
Ashok and Rita Patel Institute of Integrated Study and Research in Biotechnology and Applied Sciences, Anand, India
snehalingale@aribas.edu.in
Post Graduate Department of Bioscience, Sardar Patel University, Anand, India
mnatarajspu@gmail.com